Learning Objectives
This is a beginning level course. After completing this course, mental health professionals will be able to:
- Discuss ethical and legal considerations in providing information about medications to clients.
- Explain medication treatment algorithms for depressive disorders, anxiety disorders, ADHD, and bipolar spectrum disorders.
- Troubleshoot treatment resistant cases of depression.
- List experimental treatments for treatment resistant disorders and the data supporting them.
- Describe the newest treatment for postpartum depression.
The materials in this course are based on the most accurate information available to the author at the time of writing. New developments in the field of psychopharmacology occur each day and new research findings may emerge that supersede these course materials. This course is updated regularly as new practice guidelines are developed. This course will equip clinicians to evaluate the needs for medical treatment for their psychotherapy clients, to assess responses to treatment and to more effectively collaborate with primary care physicians and psychiatrists.
Outline
- Introduction
- Preliminary Considerations
- Legal and Ethical Issues
- Drug Metabolism and Medication Dosing
- Depression
- Introduction
- Abnormal Neurobiology: A Compelling Case for the Use of Antidepressants
- Psychopharmacology
- Bipolar Spectrum Disorders
- Introduction and Diagnostic Issues
- Treatments for Bipolar Disorder
- Anxiety Disorders
- General Considerations
- Diagnostic Issues
- Psychopharmacology
- Adjunctive Treatments
- Attention Deficit/Hyperactivity Disorder (ADHD)
- Introduction
- Diagnostic Issues
- Neurobiology
- Psychopharmacology of ADHD
- Appendix
- Psychiatric Medications: Quick Reference Guide
- Mood Stabilizers/Anti-manic agents
- Miscellaneous
- Caffeine Questionnaire
- Suggested Readings
- References: Psychopharmacology
- References: Neurobiology
- Note: “Quick Reference to Psychiatric Drugs” is available as a free download on the following website. It is typically updated once or twice a year. It may be helpful to have a copy of this as you read the following material. www.Psyd-Fx.com
Introduction
This online continuing education course is designed to teach the basics of
clinical psychopharmacology for practicing psychotherapists. The focus will
be on three groups of commonly encountered clinical conditions: mood disorders
(depression and bipolar spectrum disorders), anxiety disorders and ADHD. An
assumption is made that you are very familiar with psychopathology and DSM
diagnostic criteria, however there will be supplemental diagnostic information
presented in this course as it applies to treatment decision making. More recent epidemiological studies and new findings in the neurosciences
have influenced changes in some diagnostic criteria in the DSM-5 which will be
addressed in this course. In addition, there will be brief mention of neurobiology
and pathophysiology associated with certain clinical conditions, although a
comprehensive discussion of these topics is beyond the scope of this course
(see J. Cooper, et al. 2003 and Preston, et al., 2017 for a more detailed review).
The primary focus of this course is to provide a current overview of psychopharmacologic
treatment guidelines. Many of these are derived from large-scale empirical studies
(often referred to as algorithm studies). Treatment guidelines do not represent
personal opinions of the author, but rather are presentations of algorithms
that have been developed by NIMH supported programs, guidelines from the American
Psychiatric Association, and the Texas Medication Algorithm Project.
All
psychotherapists must be familiar with psychopharmacology for three reasons:
- It is important to know which disorders may respond to
medication treatments so that referrals can be made to an appropriate
prescriber. It is a part of informed consent to provide clients with
information regarding all viable treatment options for their particular
disorder so that they can make choices about which treatment to pursue.
- It is important to be able to communicate clearly with
physicians to whom our clients are referred to share relevant information about
the diagnosis and the client’s response to treatment.
- In these managed care days, increasing numbers of clients are
receiving psychiatric medication treatment in primary care medical settings.
Here the amount of time spent with the physician is very limited. Our
clients often want and need additional information regarding their medication
treatment that is not readily available from the primary care doctor.
Psychotherapists can be very instrumental in providing important information
regarding such issues as: side effects, how much time is required for
medications to begin working, realistic expectations for treatment outcomes
with medications, etc.
I hope that you will find this course to be helpful in your clinical practice.
Also, please see the Appendix, which offers a detailed printable guide to psychiatric medications
(doses, side effects, required lab tests, etc.); this reference may be useful
to share with clients.
Preliminary Considerations
Legal
and Ethical Issues
A review of case law reveals that there are a number of
cases in which non-physician health care professionals have been accused of
practicing medicine without a license because they gave patients information
regarding their medications and medical treatment. Obviously, it is important
for all therapists to practice within their scope of practice, to do whatever
is in the best interests of our patients and to be on solid ethical ground. It
is clear from the existing case law that it is unethical and illegal to tell
patients either: 1. stop taking a medication or 2. to change the dose of a
medication. This is considered practicing medicine. However, in every case
(with one exception addressed below) those who provided information regarding
medicines and medication treatment and were accused of practicing medicine
without a license were found to be not guilty. In half of the cases, further,
the judge said that not to provide information to patients may have been acting
in a professionally incompetent manner. Here is what this is based on. First
and foremost is the right granted by the first amendment (i.e. right to free
speech). Secondly, the following are deemed appropriate to share with patients,
if and only if the therapist has training and is knowledgeable regarding the
facts of medication treatments:
- Once the
diagnosis is made, to tell the patient about treatment options. This is a part
of “informed consent” in which the therapist must share with patients their
available treatment options (mentioning those treatments that are empirically
supported). Therefore, even if a therapist is not especially fond of pharmacological
treatments, it behooves them to share information regarding drug treatments. At
the heart of informed consent is a respect for our clients in terms of allowing
them to be the final judge in opting for certain treatments.
- Discussion
about medication side effects
- Discussion
regarding the likely or common positive benefits of drug treatments (i.e. what
kinds of symptoms are likely to be improved with medications)
- Discussions
regarding the length of time that it may take for medications to begin to show
clinical effects
- Limitations of
medication treatments
- Underscoring
the importance of collaborative treatment, e.g. the value in having both the
therapist and the prescribing doctor share information about the course of
treatment (e.g. improvement, lack of improvement, emergent side effects, etc.)
- There is one case in which a non-medical clinician was found
to be unethical and this was due to the fact that this person only recommended
an over-the-counter produce but failed to talk about other available treatment
options. As will be discussed below, some over-the-counter products do have
empirical support for efficacy in treating some psychiatric disorders, and can
be mentioned to patients as options. However, this must be done in the context
of presenting all available empirically validated treatment options.
Due to the impact of managed care, many patients are
receiving prescriptions for psychotropic medications from primary care
physicians who have very limited time to spend with the patients both initially
(when the diagnosis is made and treatment is initiated) and in follow-up. This
is a significant problem and psychotherapists can provide enormous help to
patients by monitoring their medication treatment and providing support and
information regarding drug treatments, and be able to do so in ethical and
legal ways. Ultimately the care of our patients is paramount.
Drug Metabolism and Medication Dosing
For each psychiatric
medication discussed in this text, you will see listed the typical adult daily
doses. In many instances the “therapeutic dosage range” is broad. For example,
daily dosing with lithium is between 600 and 2400 mg. or for Prozac, 10-80 mg.
per day. It is important to know that the amount of medication required to
effectively reduce and eliminate symptoms often has little to do with how
severe the symptoms are. And what matters is not so much how much drug is
ingested but rather, how much of the medication enters the blood stream.
There are three
primary factors that influence the amount of drug that finds its way into the
blood stream. First is the rate of liver metabolism. Psychotropic medications
are absorbed through the walls of the stomach and intestines and go directly to
the liver. Here the drug molecules are acted upon by liver enzymes that begin a
process generally referred to as biotransformation. Liver enzymes chemically
alter the medication in ways that allow the drug to be more readily excreted
from the body. The liver’s function is to detoxify the body. Thus, in this
so-called “first pass effect” through the liver, a good deal of the drug is chemically
transformed and then rapidly excreted from the body. However, some of the
medication initially escapes this process, makes its way through the liver and
into circulation and thus is allowed to begin accumulating in the blood stream.
How rapidly the liver metabolizes drugs depends on a number of factors. This
resulting blood level is what matters when it comes to reducing symptoms.
(Note: two psychotropic medications are not metabolized in the liver: lithium
and Neurontin).
Genes play a
significant role in this process. A small percentage of people are known as
rapid metabolizers. They take certain drugs and then eliminate them very
quickly. The result is that even though they may be taking what seems like an
adequate dose of the medication, little actually gets into the blood stream.
Once it is discovered that someone is a rapid metabolizer, and then usually
it is required that they be prescribed high doses of medications and eventually enough gets
into the blood stream to be effective. Again, this has nothing to do with how
severely ill they are … it’s just a matter of the liver’s metabolic rate.
Conversely are hypo-metabolizers. This also small percentage of people (perhaps
5% of the general population have fewer than average liver enzymes. It is important to note that those of Asian and African descent show higher percentages of hypo-metabolism: approximately 33% and thus more caution should be taken with initial dosing in these individuals to avoid significant side effects). The
effect is that they can take a “typical” starting dose of a medication, and on
its trip through the liver, only small amounts are transformed and excreted.
The result is often very high blood levels of the medication and severe side
effects or toxicity. The ultimate solution for hypo-metabolizers thus is to use
very small doses of medications initially and then increase doses gradually.
Sometimes when a person is first treated they will experience serious side
effects and this may be due to hypo-metabolizing. It is often hard to know
ahead of time if this will happen with any one given individual. Thus, if your
patient has had an experience of encountering very intense side effects with
other medications in the past, one may anticipate that they may be a
hypo-metabolizer, and thus initial dosing should be low and increased dosing should be done gradually.
A second factor determining blood levels of medications is the functioning
of the kidney. Sometimes genetic factors play a role here too, but more often
problems can occur due to kidney disease. Thus, for some bipolar medications,
in particular, pretreatment labs will include an assessment of kidney functioning
(this is especially important for patients being treated with lithium).
Finally, a number of drugs can adversely affect liver
metabolism and thus alter blood levels. Here is where drug-drug interactions
can cause significant problems. This applies to some prescription drugs,
over-the-counter drugs, herbal and dietary supplement products and recreational
drugs. The use of prescription drugs must be carefully monitored by the
treating prescriber. In addition, even modest amounts of alcohol can have
significant effects on the liver. St. John’s Wort, a popular herbal product for
the treatment of depression, is well known for causing some very significant
changes in liver metabolism.
Depression
Introduction
The World Health Organization announced findings from a multi-nation study
indicating that currently for women major depression is the second most disabling
condition (among all medical and psychiatric disorders). And by the year 2020
it will rank as the number one overall disabling condition, worldwide (males
and females combined) (Murray and Lopez, 1996). Each year 25 million Americans
suffer from serious depression, it has a lifetime prevalence rate of 17% (Kessler,
McGonagle, Zhao, et al. 1994), and ranks as the second most commonly seen disorder
in patients seeking treatment from primary care physicians. Data suggests that
the incidence of depression is increasing (Murray and Lopez, 1996). Morbidity
and mortality associated with serious mood disorders is enormous. Suicide is
the 8th leading cause of death in the United States, and chronic
and recurrent depressions significantly increase risk for coronary heart
disease, stroke, and osteoporosis (NIMH, 2003). By 2020 the World Health Organization projects that depression will be the second leading condition leading to decreased life expectancy.
It is estimated that only 50% of those in the United States who suffer from major depression seek treatment. Additionally,
treatment received is often very inadequate. In the United States most drug
treatment for depression takes place in primary care medical settings. Eighty-five
percent of prescriptions written in the United States for antidepressants are
written by physicians and nurses that do not have specialty training in psychiatry.
This is due in large part to managed care’s efforts to cut costs by having the
majority of psychiatric treatment take place in primary care settings. Treatment outcomes in primary care medical settings are poor largely due to the lack of adequate follow-up.
A Changing Treatment Landscape
The 1980’s and 1990’s saw the proliferation of HMOs and managed care companies
that attempted to streamline mental health care in order to achieve cost containment.
During this same time psychiatry training programs have increasingly emphasized
psychopharmacology as the mainstay of treatment for clinical depression. Additionally,
during the past decade the number of medical students choosing psychiatry as
a specialty has dropped by 50%. In years to come, there will be increasing unavailability
of psychiatrists, likely further contributing to more and more treatment being
conducted in primary care settings. Finally, pharmaceutical advances and marketplace
forces have had a substantial impact on shifting patterns of practice in the
mental health arena (see Figure 1).
Figure 1.
Treatment Received by Patients Diagnosed
with Major Depression* |
| |
1987 |
1997 |
Antidepressants |
37% |
75% |
Psychotherapy |
71% |
60% |
* Olfson,
Marcus, Druss, et al. 2002, based on sample size N = 32,000 |
In many settings
psychopharmacology has become the primary or sole form of treatment for many
suffering from serious mood disorders. Yet, the vast majority of patients
treated either do not respond or only experience partial responses.
Treatment Outcomes
How effective are antidepressants? All drugs must go through rigorous trials
prior to FDA approval, and must demonstrate clear superiority over placebos.
The currently available antidepressants have all passed the test, but the outcome
data is often spurious and plagued by methodological flaws. First and foremost,
in the majority of pre-FDA approval efficacy studies patients selected for medication
trials hardly resemble typical outpatient populations. Patient selection excludes
those who have had prior suicide attempts, who have psychotic or bipolar symptoms,
and who have comorbid medical illnesses, substance abuse or personality disorders.
A paper by Zimmerman, Posternak and Chelminski (2002) looked at consecutive
patients seen in an outpatient setting who were diagnosed with unipolar, major
depression. If patient selection criteria as noted above were applied to this typical group of depressed outpatients, 85% of them would be excluded
from drug trials. Complex comorbidity is the rule, not the exception in usual
clinical settings. Thus, it may not be appropriate to generalize results from
efficacy studies to the real world of clinical practice.
Another significant
flaw in the reporting of outcome data is the common practice to exclude
dropouts due to side effects from percentages of responders (Gitlin, 2002).
Thus, for most antidepressants studied, the positive outcomes are inflated (see
Figure 2).
A more appropriate
picture of outcome is derived from what are termed Intent-To-Treat (ITT)
studies. In these studies, dropouts are
considered to be failures (non-responders).
Figure 2.
Antidepressant Treatment of Major
Depression Outcome Results:
General Results Reported from Drug Efficacy
Studies |
| |
Commonly Reported
Outcomes |
ITT Outcomes |
Side-effects drop-outs |
15% |
15%* |
Responders |
60% |
50% |
Non-Responders |
40% |
35% |
* Drop-out rates due to side effects vary among new generation
antidepressants ranging from Wellbutrin, SR: 9%, to Paxil: 21% |
Upon closer inspection, the picture is even less positive than these ITT data
suggest. In most studies “responders” are those who achieve a 50% or greater
decrease in scores on the Hamilton Depression Rating Scale (Ham-D) or a Ham-D
score of 7 or less. In most studies only half of the responders reach Ham-D
scores below 14, and although they are significantly improved, they still are
not fully recovered. Thus, generally, only 25% reach full remission if they are treated with one medication. Failure to reach full remission is associated with ongoing
subclinical symptomatology (often patients report just not feeling enthusiastic
about life) and with partial responders there is a three-fold increase in acute
relapse of major depression (Paykel, Ramana, Cooper, et al., 1995). Finally,
even those judged to be in full remission, often continue to experience subtle
residual depressive symptoms. In one study only 18% of “fully remitted” patients
were truly asymptomatic (Nierenberg, Keefe, Leslie, et al., 1999).
It must be
emphasized that this critique of outcome data is not intended to be an
indictment of antidepressants. Quite
the contrary, these medications have been shown to be effective in
alleviating many serious mood disorder symptoms (e.g. vegetative symptoms),
undoubtedly have saved many lives, and antidepressants may also be
neuroprotective (see below). But they
are not a panacea. Such is the case
with most medications that are used to treat chronic illnesses such as
diabetes, hypertension, cancer, chronic pain, etc. Likewise, most studies of
psychotherapies for depression (e.g. cognitive-behavioral therapy,
interpersonal therapy, etc.) report success rates in the 50-55% range. It is
humbling to appreciate the limits of any single treatment modality.
What has become clear is that mood disorders are tremendously complex and heterogeneous
disorders involving numerous interacting variables: e.g. genetic predispositions,
life stresses, interpersonal relationships, hormonal, neurobiologic, intrapsychic,
cultural and existential features. It is the appreciation of such complexity
that has led to increased interest in integrative approaches to treatment (Preston,
2006).
Abnormal
Neurobiology: A Compelling Case for the
Use of Antidepressants
A comprehensive
review of the neurobiology of depression is beyond the scope of this paper.
However, let us consider a few important research findings that have emerged
during the past decade. Sixty percent of people experiencing a major
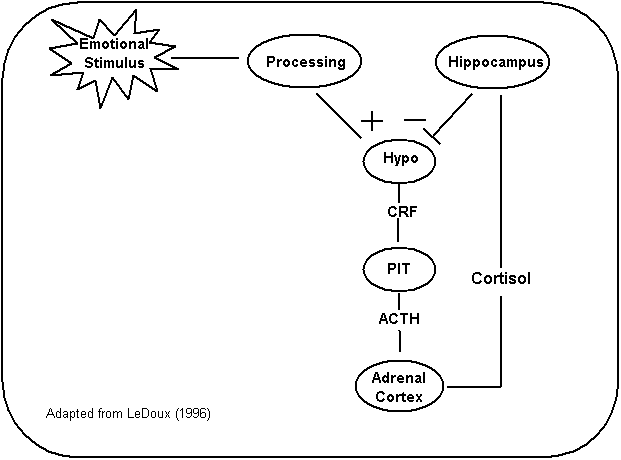
depressive episode exhibit a hyperactive hypothalamic-pituitary-adrenal (H-P-A)
neuroendocrine axis (see Figure 3).
In mammals when danger or adversity is perceived in the environment a
number of complex events take place in the nervous system, launching adaptive
fight-or-flight responses. One of the
most important components of this biological response is the activation of the
H-P-A axis, which ultimately releases glucocorticoid hormones into general
circulation (in humans to principle glucocorticoid is cortisol). Cortisol operates to facilitate the release
of glucose into the blood stream and to increase blood pressure (both of which
are essential for mounting an effective fight-or-flight response).
Figure
3.
Hypothalamic-Pituitary-Adrenal Neuroendocrine
Axis
See also, Le Doux (2015)
Cortisol enters
circulation and is distributed throughout the body. In the brain the most prominent site for cortisol response is the
limbic brain structure, the hippocampus. In adaptive fight-or-flight responses
cortisol activates the hippocampus to inhibit the H-P-A axis (operating like a
“brake”). Of course, if danger persists
in the environment, the H-P-A axis is continuously reactivated. However, when stressors subside, the
cortisol-mediated feedback loop shuts down the system, and thus plays a role in
affect regulation (LeDoux, 2017).
This hyperactivity
of the H-P-A axis, seen in many cases of major depression, results in a
condition known as hyper-cortisolemia.
Here, cortisol levels are significantly elevated and have been shown to
be neurotoxic. These high, sustained,
toxic levels of cortisol have a marked impact on hippocampal nerve cells (e.g.
cause dendrite retraction, cell death, and hippocampal atrophy). Hypercortisol
also may damage other neural tissue (e.g. anterior cingulate, amygdala,
cerebellum: all of which have been implicated in playing a role in affect
regulation). Over time the result is progressive
brain damage that likely accounts for the deteriorating course seen in many
cases of untreated or poorly treated recurrent and chronic depression and
bipolar illness (Sapolsky, 1996). High
cortisol levels in depressed pregnant women cross the placental-blood barrier
and may adversely affect the fetus’s nervous system.
Additionally,
depression reduces the gene expression of brain-derived neurotropic factor
(BDNF), which is a neuroprotective molecule known to facilitate the repair of
damaged nerve cells and to promote neuron regeneration (i.e. neurogenesis) in
the hippocampus.
Antidepressant
medications reduce cortisol levels (Heim and Nemeroff, 2002; Nestler, Hyman,
and Malenka, 2001) and reactivate the production of BDNF, (note: lithium, Depakote, Tegretol, Lamictal, Seroquel, and Equatro, and
regular exercise also increase the levels of BDNF) which can lead not only to
clinical improvement, but also to the birth of new nerve cells in the
hippocampus (Kempermann and Gage, 1999). In this way antidepressants can play a
crucial role in treating symptoms of depression as well as protecting the brain
from the effects of toxic levels of stress hormones.
Diagnostic Issues
For many years a
distinction was made between so-called “reactive depressions” or “psychological depressions” (by definition,
depressions that emerged in the wake of significant psychosocial stressors)
and “endogenous depressions”
(presumably due to the effects of certain changes in neurobiology…e.g.
genetically based mood disorders, secondary to various medical illnesses such
as thyroid disease, or due to the impact of substances, e.g. alcohol,
antihypertensive drugs). Currently the distinction between endogenous and
reactive depressions is less relevant. The critical diagnostic issues (as this
relates to the decision to treat with antidepressants) include the following:
- Marked dysfunction (social, occupational, etc.) that lasts for more
than 2 weeks or fails to respond adequately to psychotherapy.
- Patients, who are of below-average intelligence, are significantly
non-psychologically minded (i.e. unable to introspect), who refuse
psychotherapy, where professional treatment is not available, or who are too impaired to productively participate in
psychotherapy.
- The presence of neurovegetative symptoms (if these are sustained; i.e.
present most days):
- Sleep
disturbances (especially early morning awakening and middle insomnia)
- Loss of libido
- Anhedonia or a
non-reactive mood
- Appetite and
weight changes (either increased or decreased)
- Marked fatigue
- Psychomotor
retardation or agitation
- Diurnal
variations in mood and cognitive ability: generally, more pronounced in the
morning
- 66% of patients with
dysthymia (Persistent Depressive Disorder: new name introduced in DSM-5) have been shown to be responders to antidepressants and thus should
also be considered for such treatment.
- Depressions caused by a general medical condition or prescription
medications. Typically, the treatment strategy is to focus treatment on the
general medical condition directly and only use antidepressants if such
treatment is unable to resolve the depression. Likewise, a change in
prescription medications (e.g. changing the type of antihypertensive medication
used to treat high blood pressure to a different class of antihypertensive) may
be effective in reducing depressive symptoms. However, if not the addition of
an antidepressant may be considered.
Psychopharmacology
Antidepressant Medications
Antidepressants and daily adult dose ranges (only the newer generation antidepressants,
which are in common use, are listed below). These medications have been developed
since the late 1980s and are considerably safer and easier to tolerate than
older-generation tricyclic and MAOI antidepressants
Generic Name |
Brand Name |
Typical Adult Daily Dose |
fluoxetine |
Prozac, Sarafem |
10-80 mg |
bupropion |
Wellbutrin |
150-400 mg |
sertraline |
Zoloft |
50-200 mg |
paroxetine |
Paxil |
20-50 mg |
venlafaxine |
Effexor |
75-350 mg |
nefazodone |
Generic only |
100-500 mg |
mirtazapine |
Remeron |
15-45 mg |
citalopram |
Celexa |
10-60 mg |
escitalopram |
Lexapro |
5-20 mg |
duloxetine |
Cymbalta |
20-80 mg |
atomoxetine |
Strattera |
60-120 mg |
| desvenlafaxine |
Pristiq |
50-100 mg |
| vilazodone |
Viibryd |
10-40 mg |
| vortioxetine |
Trintellix |
10-20 mg |
| levomilnacipran |
Fetzima |
40-120 mg |
Pharmacologic Treatment Guidelines
During the past 20 years large-scaled empirical studies have been designed in order
to establish guidelines for what is currently referred to as “evidence-based
medicine”. These include the STAR-D (Sequenced Treatment Alternatives for Relieving
Depression: a multi-site study sponsored by the National Institute of Mental
Health), the Texas Medication Algorithm Project and UCLA’s Targeted Treatment
for Depression Program (Metzner, 2000). Treatment guidelines in psychiatry have
been influenced by three factors: 1. Marketplace variables (e.g. pharmaceutical
company advertising; decisions made by HMOs and managed care companies to adopt cost-effective drug formalities),
2. Consensus (i.e. invite “experts” to convene a panel and discuss pros and
cons of various medical treatments and agree upon best treatment strategies),
and 3. results from a large amount of research regarding treatment outcomes.
The latter of these offer important information, but may be inherently flawed
because most psychopharmacology outcome studies are sponsored by drug companies
who have a vested interest in producing good outcomes. The major drawback here
is that many null studies never make it into the journals. Thus, it is difficult
to realistically evaluate the effectiveness of certain treatments with only primarily
positive outcome data available.
The large-scaled
studies mentioned above are funded by the NIMH, Texas Department of Mental
Health, and a university, respectively, and thus may more accurately reflect
realistic outcome data, non-influenced by the profit motives of drug
companies. Also, the studies are not
ones of consensus opinions, but rather based on empirical outcomes with very
large groups of subjects.
What is summarized
below are the first of what promises to be a growing body of evidence-based data
that can suggest specific strategies for the treatment of major depression.
First-Line Medication
Choices: Major Depression
(Metzner, 2000;
Shelton and Tomarken, 2001; Goodwin and Jamison, 2007).
Major depression as
defined by DSM-5 criteria (APA, 2013) represents a heterogeneous group of mood
disorders that vary in terms of severity (mild-to-severe), clinical/symptomatic
presentation and presumed etiology. A
large body of neuroscience research has strongly implicated that dysregulation
of certain central neurotransmitters may be associated with particular
psychiatric symptoms. Most individuals that experience a decreased availability
of neurotransmitters such as serotonin (abbreviated 5-HT) dopamine (DA) or
norepinephrine (NE) do not develop clinical depression (Delgado, Charney,
Price, et al., 1990). However, some do,
which is likely due to underlying genetic or other vulnerability factors. Among depressed subjects inadequate
functioning of 5-HT, DA, or NE can contribute to certain core depressive
symptoms such as a depressed mood, pessimistic and negative thinking, guilt,
low self-esteem, and fatigue. What has emerged during the past two decades of
research are general but consistent findings suggesting that beyond common,
core symptoms of depression particular neurotransmitter dysfunctions may be
accompanied by or cause specific symptoms:
- Serotonin
Dysregulation: irritability,
impulsivity, suicidal impulses/behavior, ruminations
- Dopamine and/or
Norepinephrine Dysregulation: apathy,
anhedonia, low motivation
Closely
paralleling these findings from neuroscience research are data from empirical
pharmacologic studies (e.g. Metzner, 2000), which have led to the following
guidelines in which particular symptomatic features point toward first-line
antidepressant medication choices:
Unipolar Major Depressive Disorder (MDD)
Treatment Algorithm: Choosing a First-Line Antidepressant
- Major Depression with agitation and/or comorbid anxiety (represents
about 50% of cases of major depression). Medications recommended: Selective
Serotonin Reuptake Inhibitors (SSRIs: Prozac, Zoloft, Paxil, Luvox, Celexa,
Lexapro, Viibryd, Brintellix). It is likely that SNRIs (serotonin and norepinephrine reuptake inhibitors such as
Effexor, Pristiq, Fetzima, and Cymbalta) may also be useful in this group of depressed
patients, however, this has not been adequately studied as of yet. One action seen with acute administration of SNRIs (i.e. within a few hours after taking the first dose) is to increase sympathetic nervous system activity (by increasing the availability of norepinephrine in the sympathetic system) and this can be experienced as increased anxiety within a few hours after taking the first dose. For this reason, SSRIs are preferred over SNRIs in treating major depression with comorbid anxiety. Additionally, patients that present with irritability, impulsivity,
rumination and/or significant suicidal ideations may also preferentially respond
to these serotonin-specific antidepressants. Although these agents may prove
to be effective in treating agitated/anxious depressions, a significant problem
is commonly encountered as treatment begins. This is a side effect referred
to as activation (affecting approximately 10% of patients); which is experienced as increased energy, anxiety,
initial insomnia, and/or agitation typically emerging several hours after
taking the first dose (also this can occur shortly after dose escalations). In individuals with this version of MDD, such acute
onset side effects often result in abrupt patient-initiated discontinuation
(the side effects often scare patients and may make them very reluctant to
go through another antidepressant trial). Thus although after 4-6 weeks of
treatment SSRIs often begin to significantly reduce both depression and anxiety
symptoms, the initial few weeks of treatment can be very challenging and many
patients drop out of treatment. A popular and effective way to address this
problem is to initially co-administer a benzodiazepine (i.e. minor tranquilizer,
e.g. Ativan), using this drug to rapidly reduce anxiety, agitation, and drug-induced
activation. In addition, since benzodiazepines begin to reduce anxiety within
an hour of taking the medication, the experience for many patients is that
they feel noticeably better, quickly. This is an added benefit because it
often leads to hopefulness about treatment and enhances compliance. Generally,
after one month of treatment the benzodiazepine can be phased out. This is
often a very successful strategy, however there is one important warning.
Since benzodiazepines can be drugs of abuse, the use of these agents is risky
and not indicated if there is a history of alcohol or substance abuse. With
an addiction history, some psychiatrists are now using the non-habit-forming
antihistamine hydroxyzine (Atarax; Vistaril) to target the activation and Benadryl or low doses of trazodone (25-50 mg) or low dose Remeron (7.5-15 mg) for initial insomnia.
- Major Depression with marked anhedonia, apathy, and/or amotivation (represents about 30-35% of MDDs). Such depressions are sometimes referred to as anergic depressions. The recommended antidepressants
are Wellbutrin (that targets DA and NE) and norepinephrine reuptake inhibitors
(NRIs) e.g. Strattera (note: Strattera is FDA approved for treating ADHD but is, in fact, a norepinephrine reuptake inhibitor antidepressant.) New NRIs are used in Europe and are likely to be released on the USA market in the near future.
- Atypical Depression (15-20% of MDDs). Atypical symptoms include:
hypersomnia, appetite increase, carbohydrate craving and subsequent weight
gain, and severe fatigue. Other symptoms often associated with atypical MDD
are: rejection sensitivity and anxiety attacks. Medications of choice are
SSRIs, monoamine oxidase inhibitors (MAOIs) and possibly Wellbutrin (APA,
2003). Note that Wellbutrin may increase anxiety; thus, if anxiety symptoms
are present, this drug may be contraindicated. It is important to exercise
caution when treating atypical MDD because many such individuals may be expressing
the depressive phase of bipolar illness, and if treated with antidepressants
alone there is a risk of switching (i.e. provoking mania or hypomania) or
cycle acceleration (the tendency to cause increasingly severe or more frequent
mood episodes). It has been found that 80% of those suffering from atypical
depression ultimately are discovered to have bipolar disorder (usually, bipolar
II). More will be said about switching and cycle acceleration in the section
below on bipolar illness.
- Very Severe and/or Highly Recurrent Cases (symptoms can be anxious,
anergic, or atypical) These more severe and recurrent disorders appear to
respond best to dual action antidepressants such as Effexor (NE and 5-HT),
Cymbalta (NE and 5-HT), Pristiq (NE and 5-HT), Remeron (NE and 5-HT), Trintellix (5-HT and DA), or the
combination of an SSRI and Wellbutrin (this combination actually becomes triple
action, i.e. targeting NE, DA, and 5-HT).
Esketamine and Beyond:The drug ketamine has been used in medicine and veterinary medicine for years as an anesthetic. It also has been abused as a street drug, because of its dissociative and hallucinogenic properties (on the street often referred to as Special K). It is also widely used in psychiatry mainly in ketamine clinics to treat severe, treatment refractory depression and especially acute, severe suicide risk. In psychiatry it is used off label and administered typically twice a week in the clinic, because it requires IV dosing and a period of 2-3 hours of patient observation afterwards. This is because dissociative and some psychotic symptoms exist for a period of several hours after dosing. Some severely affected patients respond rapidly to the antidepressant effects. Also, there is often a rapid decrease in suicidal ideation and impulses. It has also been used successfully to treat patients with psychotic depressive episodes.
Recently the active S isomer of Ketamine (esketamine; brand name Spravato) has been FDA approved to treat depression and acute suicidal risk. It is available in a nasal spray. Because of its abuse potential, it is tightly regulated by both pharmacies and treatment facilities. It must be administered in the treatment facility. The current cost ranges from $4500-$6500 per month, but because it is FDA approved, it may be covered by insurance. The duration of effects and even the frequency of dosing is largely unknown. S-Ketamine is a glutamate NMDA antagonist, although its specific mechanism of action is largely unknown. In many individuals, however, the effects are seen within minutes to hours. The success of S-Ketamine opens the door for future drug development because its action on glutamate is clearly different than other, standard antidepressants.
Metzner (2000) has
demonstrated that targeted treatment for depression (i.e. TTD) where the
choice of a first-line antidepressant is based on the presenting clinical
picture (as outline above) yields superior outcomes compared to standard (STD)
treatment as usual. Positive responses to
antidepressants: STD = 65%, TTD = 96%. (It is important to note that these figures represent “responders” and not necessarily those reaching full remission).
Partial and
Non-Responder Strategies
Fifty-five to sixty-five percent of patients treated with antidepressants only
experience a partial response or no response at all when receiving their first antidepressant trial (Paykel, Ramana, Cooper,
et al., 1995; Doraiswamy, Kahan, Donahue and Richard, 2001; NIMH: STAR-D,
2008). And as noted earlier, those who do not reach full remission incur an
increased risk of relapse (Paykel, Ramana, Cooper, et al., 1995). Empirical
studies are beginning to provide databased treatment guidelines for partial
or non-responders (see Texas Department of Mental Health, 2005 and NIMH: STAR-D
Program, 2008).
It must first be recognized that a number of factors may
account for less than adequate antidepressant responses, including the
following:
- Poor adherence
- Excessive use of caffeine or energy drinks that can significantly interfere with slow wave (deep) sleep which can aggravate depression
- Unreported
substance abuse (in particular, alcohol use/abuse which may both exacerbate
depressive symptoms and interfere with the metabolism of antidepressant
medications)
- Unsuspected
medical illnesses (e.g. sleep apnea, restless legs syndrome, or thyroid disease)
- Failure to
address key psychological conflicts and/or family based psychopathology
- Choice of wrong
class of antidepressant medication (e.g. pharmacologically targeting the
serotonin system when in fact a particular patient’s depression is associated
with dysregulation of norepinephrine)
- Chronic residual
brain impairment sustained during previous untreated or inadequately treated
depressive episodes (due to hypercortisolemia)
Partial Responder Strategies: (Trivedi and Klieber, 2001): the highest
yield next step strategy is to progressively increase the medication dose. This was also born out in the STAR-D program that demonstrated better outcomes for very ill and treatment resistant cases by using high doses of antidepressants.
Some patients who are hyper-or rapid metabolizers require higher doses to achieve
adequate blood levels. The doses can be progressively increased if tolerated.
If this strategy is ineffective or impossible owing to emergent side effects,
step two is to augment (i.e. to add an additional medication to the current
drug). Augmentation strategies often yield good responses in 35-65% of those
treated. The following are common augmentation strategies that often are successful:
- SSRI plus Wellbutrin
- SSRI plus
stimulant (e.g. Ritalin, once a day, morning dose) for anergic depressions
- Antidepressant
plus lithium (generally lithium levels of 0.4 – 0.6 mEq/l)
- Combining
Effexor and Remeron (Stahl, 2000)
- SSRI plus atypical antipsychotic (e.g. Zyprexa, , Seroquel, or Abilify). Note: this strategy is often successful in treating
partial responders who have major depression without psychotic features. Abilify is FDA approved for augmenting antidepressants in the treatment of major depression.
- Antidepressant
plus T3 (triiodothyronine), 25-50
micrograms per day
Non-Responder Strategies (Trivedi and Klieber 2001): Once again one
must evaluate issues involving adherence and possible substance abuse. Should
these issues be ruled out, then the highest yield next step strategy is to optimize
the dose (dosage increases, if tolerated). Should a high dose be reached and
there is still little or no response, then the next step strategy is to change
classes of medications (e.g. switch from a serotonin active drug [SSRI], e.g.
Zoloft, to an antidepressant that targets norepinephrine and
dopamine [e.g. Wellbutrin]).
Phases of Treatment
There is general agreement among
research groups that the treatment of major depression involves three phases
(American Psychiatric Association, 2003; Texas Department of Mental Health,
2003):
Acute Phase: Starts with the first dose and extends until the patient
is asymptomatic. Since symptoms have abated, many clients will naturally think
that they no longer need medications and will discontinue (against medical advice).
At this point in treatment should patients discontinue, more than one-half will
experience an acute relapse within a few weeks (Stahl, 2013). Ongoing antidepressant
treatment, however, decreases the likelihood of acute relapse, necessitating
the next phase:
Continuation Phase: Continue treatment at the same dose for a
minimum of 6 months. If during this time period there are no breakthrough depressive
symptoms, then discontinuation can be considered (gradual discontinuation, e.g.
over a period of six weeks is strongly recommended to avoid discontinuation
withdrawal symptoms).
Maintenance Treatment: Lifelong treatment is strongly indicated for
patients with highly recurrent major depressions (e.g. those in their 3rd or subsequent episodes). Chronic treatment attempts to prevent recurrence and can be helpful in this regard.
Treatment of Depressive Sub-types
Minor depression: Patients suffering from Persistent Depressive Disorder (dysthymia) have shown
a very positive response to antidepressant medication treatments (33% good
response, 33% very good response) (Akiskal and Cassano, 1997).
Seasonal depression: Often responds to high-intensity light therapy, generally provided by the use of a commercially available light box or going out-of-doors without sunglasses. The typical “dose” of light therapy is 10-30 minutes of exposure to a light source that emits a minimum of 10,000 lux. The light has an impact by striking the retina, which activates a specific nerve pathway (the retinal-hypothalamic nerve). In most cases, high-intensity light therapy must be accompanied by the use of antidepressant medications (Rosenthal,, 2013). Note that some seasonal mood disorders are associated with bipolar illness and thus one must exercise caution in using bright light therapy to prevent a shift into mania.
Pre-Menstrual Dysphoric Disorder (PMDD). If mood symptoms are
only seen for discrete periods of time prior to menstruation and absent the
rest of the month, acute treatment with SSRIs is often successful (while all
other mood disorders require 2-6 weeks of treatment prior to the first signs of
clinical improvement, many of those suffering from PMDD realize symptomatic
relief a few hours after taking the first dose of an SSRI). Often this strategy allows the PMDD patient
to avoid chronic medication use (i.e. needing to take the medication only
during those days each month when mood symptoms are present). (Nutt, 2002).
Treatment of Psychotic Depressions
Success
rates for treating psychotic disorders with single agents are generally poor
(American Psychiatric Association, 2003). For this reason the combined use of
antidepressants and antipsychotics is required.
Phases
of treatment for psychotic depression recommend a period of one-year
continuation treatment with both antidepressants and antipsychotics before a
trial discontinuation. Premature
discontinuation is associated with a high risk of acute relapse. And poorly treated psychotic depressions
carry a higher risk of suicide than do other forms of MDD. Should pharmacologic treatment fail, ECT
(electroconvulsive/shock therapy) is often highly effective.
Highly
Treatment Resistant Cases
If despite standard treatments or augmentation strategies there is still little
or no response, be sure to reassess for the presence of:
- Unreported substance abuse
- Undiagnosed primary sleep disorders, e.g. sleep apnea or restless legs (these disorders frequently cause depressions that fail to respond to standard antidepressant treatments)
- Unsuspected
occult illnesses (e.g. endocrine disease, cancer, etc.). Subclinical hypothyroid is often a cause for antidepressant failures or partial responses. TSH levels, even in the upper part of the “normal range”, i.e. 2.0-3.0, may produce inadequate antidepressant responses. Ideally those who are clinically depressed need TSH levels around 1.0, and for those with slight elevations in TSH often respond to treatment with very doses of T3 or T4)
- Undiagnosed bipolar disorder (bipolar depressions are often especially difficult
to treat and treatment with antidepressants at times can aggravate the disorder causing
switching and/or cycle acceleration). Additionally, a common result is that antidepressants simply fail to produce antidepressant responses if the patient, in fact, has bipolar disorder.
- Other possible
factors:
- Secondary gain
- Unaddressed psychological issues (e.g.
internal conflicts or unmourned losses)
- Psychologically toxic interpersonal
environment that has not been addressed (e.g. no intimacy, emotional abuse,
domestic violence).
Postpartum Depression
Clinically significant postpartum depression can affect up to 10-14% of women giving birth. Technically, this can occur during pregnancy but more commonly occurs after the birth of the infant. Especially if there is a pre-existing depression, the onset can follow rapidly after delivery. However, more commonly, the average onset is 6 weeks following delivery. Postpartum depression is distinct from what often is referred to as “the baby blues”. Such a condition is not pathological but rather reflects increased emotional sensitivity in general (for the full range of emotions, including happiness as well as bouts of sadness or tearfulness). It is thought that this phenomenon, common to most new mothers, is adaptive in its contribution to increased empathy, nurturing and mother child bonding. Postpartum depression is quite different.
Postpartum depression often manifests as moderate to extremely severe depression. It’s typical onset around 6 weeks has raised questions about the adverse impact of sleep deprivation, especially during the first weeks or months following delivery. Technically, severe depression with onset during the first year after giving birth to a baby may be a manifestation of postpartum depression. This condition, beyond causing significant suffering for the mother, can be quite severe and often quite risky. Many women experiencing a severe episode are completely unable to hold or disinterested in holding their babies. This can profoundly interfere with critical early child-mother bonding (consequences that may have a life-long impact). The greatest risk is postpartum psychosis. Here, mothers do not interact at all with their children and in many cases they include the child in their delusional thinking. Estimates show that each year in the United States 150 babies are killed by their mothers in the midst of florid psychotic thinking and behavior. All women with postpartum psychosis should be strongly considered for hospitalization.
The standard in practice for many years is the combined use of antidepressants and antipsychotics (especially if psychotic symptoms are present). Response rates are slow and such depressive and psychotic symptoms (if untreated) last longer than most unipolar depressive episodes. They may continue for 1.5 or more years or before spontaneous remission. Standard treatment requires at least 2-4 weeks to begin to respond.
Brexanolone (brand name Zulresso) is a new drug with unique actions. It requires gradual IV infusion over a period of 60 hours and must be administered in the hospital. This is the first FDA approved drug for postpartum depression. It is presumed to have actions on allopregnanolone, a neuro-steroid that has actions on the GABA system. Both with premenstrual depression and postpartum depressions allopregnanolone levels drop precipitously. Progesterone may be targeted by the drug but the precise mechanism of action is unknown at the time of this course update. Typically, 30% of women experience a significant and rapid reduction in psychiatric symptoms. The drug may have enduring positive effects for at least a month, but longer-term follow-up studies have not been done to date. Also, most patients must be co-treated with antidepressants and possibly antipsychotics during and after brexanolone treatment. The cost of the medication alone is currently $34,000 which does not include the cost of the hospital stay and the clinicians involved. Since it is FDA approved, it may be reimbursed by some insurance companies. The cost is very high, however, this condition should be considered potentially life threatening which may justify the cost. The most common side effect is dizziness. Like all new drugs with totally unique action, such drug development may pave the way for development of other compounds. A pill form is currently under investigation.
Experimental Treatments
A number of experimental treatments have emerged during the
past ten years, including: repetitive transcranial magnetic stimulation
(George, 2003), vagus nerve stimulation, omega-3 fatty acid supplementation
(Peet and Horrobin, 2002), exercise therapy (Lawlor, and Hopker, 2002), SAM-e,
and St. John’s Wort. The newest in the field is oxytocin.
Repetitive Trans-Cranial Magnetic Stimulation (rTMS): is a technology that uses a powerful electro
magnet, which is able to stimulate the brain. Treatments generally last from
10-20 minutes during which an electro magnet is placed next to the left frontal
part of the skull. During the treatment, approximately 1000 surges of powerful
magnetic energy are delivered which penetrates the skull affecting metabolic
functioning in the brain tissue in the underlying left frontal lobe. This
treatment causes virtually no side effects (the exception is that about 1.5% of
people experience a seizure). There is no loss of consciousness. Like ECT,
treatments are given three times a week for 3-4 weeks. The literature on rTMS
suggests that it is a rapid and effective treatment for some cases of severe
depression. As with ECT acute relapses do occur after the rTMS treatments stop
and thus people are concurrently treated with mood stabilizers or
antidepressants to avoid relapse. This is a promising new approach and although FDA approved, it is
still considered to be experimental.
Vagus Nerve
Stimulation: is a technique that was initially developed to treat some
forms of severe epilepsy. It has been found to be effective in successfully
treating about 50-60% of people who suffer from highly treatment-resistant
depressions. A pacemaker-like device is implanted in the chest wall (beneath
the collar bone) and a wire extends up into the neck where it is wrapped around
the vagus nerve. Periodically a mild electrical stimulation is delivered to the
vagus nerve, which causes nerve activity that enters the brain. This also
appears to be a promising new treatment for some cases of severe depression.
Omega-3 Fatty Acids:
are dietary supplements that have some research support as effective
agents in reducing the severity of bipolar mood swings and major depression
(when added to standard medication treatments (Stoll, et al. 1999). The responses in bipolar patients, however have been disappointingly minimal, while a number of studies do show positive responses when used in unipolar depression. Doses that are used to treat or augment treatment of depression are 1000 to 2000 mg per day (of EPA: the specific type of omega 3 that has an impact on mood). Fish oil sources of omega 3 have greater bioavailability in the brain and are more effective than omega 3 from flax seed or walnut oil. The use of
Omega-3 fatty acids is described in more detail in the section below on the
treatment of bipolar disorder.
SAM-e (S-adenosylmethionine): is a naturally occurring bio-molecule
found in most living cells. It is felt to be necessary for carrying out a number
of important intracellular chemical reactions. SAM-e has been used in Europe
for more than 20 years as a treatment for depression. A number of studies have
shown it to be equally effective when compared to prescription antidepressants.
Most notable is the virtual lack of side effects. SAM-e may be useful in treating
bipolar depression, however one must exercise caution because it has, in fact,
been found to switch people with bipolar depression into states of mania. Doses
for the treatment of depression range from 400-1600 mg per day, although recent
investigations indicate that often the higher doses (1200-1600 mg per day) may
be necessary for effectively reducing depressive symptoms. This is available
over-the-counter (i.e. not requiring a prescription). It is recommended that
if your patient is considering using SAM-e, it should only be taken with close
observation by their treating mental health professional.
St. John’s Wort: is an over-the-counter dietary supplement that has
been found to have antidepressant properties. A meta-analysis of 22 studies has shown
that St. John’s Wort is equally effective to prescription antidepressants in
the treatment of mild-to-moderate depression (Lined, et al. 2008). This herbal
remedy is generally well tolerated with few if any side effects. There are reported
cases of possible infertility problems associated with its use, although it
is as yet unclear whether this is a common side effect. St. John’s Wort requires
daily dosing of 900-1800 mg. per day (taken in three divided doses), and typically
the first signs of symptom improvement take about six weeks to emerge. Thus,
the onset of action is somewhat longer than that seen with prescription antidepressants.
And as with any other treatment that has antidepressant properties, St. John’s
Wort can potentially provoke mania in people with bipolar illness. Caution:
although St. John’s Wort, when it is the only medication being taken, appears
to be quite safe, it has been found to cause some very significant drug-drug interactions.
It is strongly advised that patients never take St. John’s Wort without first
consulting with their physician or pharmacist.
Oxytocin: is a hormone normally produced in the hypothalamus and released by the posterior pituitary. It has undergone some limited study recently for the treatment of psychosis but is still considered an experimental drug in psychiatry.
Combined Psychotherapy and Pharmacotherapy
Aside from its role as a primary treatment for some types of
depression, a number of studies have demonstrated that psychotherapy can
enhance treatment outcomes when combined with drug treatment (e.g. McCollough,
2000) and may contribute significantly to aiding in relapse prevention
(Reynolds, Frank, Perel, et al., 1999; Evans, Hollan, DeRubelis, et al., 1992;
Thase, Greenhouse, Frank, et al., 1997). Several authors including MacFarlane
(2003) and Whisman & Uebelacker (1999) advocate treatment models that
combine couple therapy with individual and pharmacological interventions for a
more integrated treatment approach. Additionally, the psychotherapist is in the
best position for closely monitoring adherence, side effect problems, and
clinical response to medication treatment. This is especially important if the
client is being treated in a primary care setting where the therapist can
collaborate with the physician in order to optimize treatment outcomes.
Antidepressants and Increased Risk for Suicide |
|
There has been a good deal of media attention regarding potential risks of
antidepressants and increased suicidality (especially in children and
adolescents). The initial concern came from studies in England that raised
concerns about increased suicidality in young patients treated with the
antidepressant Paxil. In this study, which included 1300 patients, Paxil
was compared to placebo and reports of increased suicidality were seen
in 1.2% of placebo and 3.4% of Paxil treated subjects. This difference
is statistically significant. It is important to note that there were
no actual suicides in this group of youngsters. A problem is that “suicidality”
has been very loosely defined in this and other studies. Most times it includes: reports of increased thoughts about suicide, suicide gestures, non-lethal-intent, tension reducing self-harm (as is often seen in borderline personality
disorders), and in one instance a report of a child slapping herself.
(Brown University, 2004). Of course, actual suicides and lethal attempts
are also included under this umbrella of suicidality. The FDA has responded
to concerns about increased suicidality by requiring drug companies to
issue warnings about the use of these drugs with younger clients. Since the media blitz regarding antidepressants and suicidality
in youngsters, prescriptions written for children and teenagers have decreased
by 30-40%. There has been a corresponding increase in reported suicidal ideations among depressed youngsters, but not an increase in actual suicides. The FDA has reviewed a number of studies focusing on the use of antidepressants in youth. The total number of subjects in these studies is 4400. The risk of treatment emergent suicidal events is approximately 2% in those treated with either no medications or placebo and 4% of those treated with antidepressants. The most common “suicidal event” is suicidal ideations. In the group of 4400 subjects there were no actual suicides.
What is clear is that untreated major depression carries extreme risks of potential suicide, antidepressants take several weeks of treatment before the first signs of clinical improvement, depression can worsen during this startup period of treatment. This can happen in both drug treatment as well as psychotherapy, and thus therapists must be watchful for treatment emergent suicidality regardless of treatment used. Antidepressants can cause an acute increase in anxiety and agitation during the first 10 days of treatment (i.e., activation: affecting about 10% of adults treated and 10-15% of children and young adolescents treated) which could contribute to increased dysphoria. Maybe more importantly is that among teenagers presenting with major depression, 40% turn out to have bipolar disorder. This is true for 50% of pre-adolescent children with major depression. Antidepressants are known to pose risks for precipitating mania in bipolar patients. In younger people mixed mania is very common in bipolar disorder and dysphoric mania is accompanied by significant suicide risk, Thus, in treating major depression it is very important to make sure the depression is not associated with bipolar disorder before prescribing antidepressants. Finally, it is always important to differentiate between media hype and scientific data. |
Bipolar Spectrum Disorders
Introduction and
Diagnostic Issues
Bipolar disorder is a common type of mood disorder affecting between 3.5-6%
of the population (lifetime prevalence: Akiskal, et al. 2000). Previously it
was thought that the lifetime prevalence was 1-1.5% of the population, however,
more recent epidemiological studies and new, refined diagnostic criteria have
revealed the larger prevalence rate. It is now appreciated that there are a
number of different types of bipolar disorder and together these are often referred
to as bipolar spectrum disorders. Bipolar disorders are a group of genetically
transmitted illnesses that result in recurring episodes of depression and mania
or hypomania (see below). This is a lifelong disorder, which requires ongoing
medical treatment. Mood-stabilizing medications can effectively reduce episode
severity and frequency; however, there is currently no cure.
In adolescents and adults, sixty percent of manic episodes are classic manias (euphoric mania), 40% are referred to as mixed mania (see description below).
Symptoms of Classic Mania
- Euphoria or an
inflated sense of self-worth
- Restlessness, agitation,
hyperactivity
- High level of
energy
- Decreased need
for sleep (e.g. sleeping 3-4 hours per night, yet without daytime fatigue)
- Racing thoughts
and rapid, pressured speech
- Poor judgment
and impulsive behavior, e.g. spending enormous amounts of money, driving
fast/recklessly, marked alcohol or drug abuse, promiscuity and engaging in
unsafe sex
- Psychotic
symptoms can occur
Symptoms of Mania with Mixed Features (DSM-5; also, often referred to as dysphoric mania)
- Symptoms in
common with classic mania
- Agitation, restlessness, hyperactivity
- Decreased need for
sleep
- Racing thoughts
and rapid, pressured speech
- Psychotic
symptoms can occur
- Marked
irritability
- Negative,
pessimistic thinking
- Feelings of
worthlessness
- Suicidal ideas
Hypomania is a
milder version of mania that typically involves much less intense mood
symptoms. The duration of hypomania is often only 2-4 days and is frequently
not noticed as being a sign of illness by the person experiencing hypomania
(although most times family members are more clearly aware of the mood changes
and increased energy). During some hypomanias the person can feel highly
motivated and productive, is witty, gregarious and “upbeat” (although there is
often underlying irritability). One very common sign of hypomania is a decreased need for sleep with no daytime fatigue. DSM-5 requires a minimum of 4 days of hypomania to diagnose bipolar II disorder, however some epidemiological studies have shown that the most common presentation of hypomanias is 2-3 days (see Goodwin and Jamison, 2007). This finding lead the DSM-5 task force to include a new diagnosis: Depression with short-duration hypomania, requiring 2 or 3 days of hypomania. Among most experts, this is not seen as a separate diagnosis, but simply another version of bipolar II disorder.
There are five subtypes
of bipolar disorder:
- Bipolar I: severe manic (classic or dysphoric) and
depressive episodes (often with periods of normal/euthymic mood between
episodes).
- Bipolar
II (including Depression with short duration hypomanias): characterized by frequent, severe
and prolonged depressions with periodic, brief episodes of hypomania, a
normal/euthymic mood can occur between episodes, but often during these
in-between times there is a low-grade/mildly depressed and/or irritable mood. Judd, et al.
(2003) studied the course of bipolar II disorder for a period of ten years and
found that 15% of days were spent in major depressions, 40% in minor
depressions and only 1.4% in hypomanias.
- Bipolar III:
severe, highly recurrent unipolar depressions with no manias or hypomanias.
However, if the patient is treated with certain drugs (e.g. stimulants,
antidepressants or steroids) there is a likelihood that the medication will
provoke a switch into mania or hypomania, and from that point on, manic
episodes may begin to occur spontaneously. A more common outcome is simply that standard
antidepressant medications may be ineffective unless combined with a mood
stabilizer. This subtype of bipolar is fairly rare, and is also characterized
by early onset depressions (in childhood or early adolescence) and a positive
family history of bipolar disorder (note: Bipolar III is rare and not an official DSM-5 diagnosis, but is a diagnosis worth considering in cases of treatment resistant depression. They may respond to mood stabilizers).
- Pre-pubertal or
early adolescent bipolar disorder: likely to be a more severe variant of
bipolar disorder. Manias tend to be non-episodic (i.e. more chronic) and 95%
are of the mixed/dysphoric variety. Note: In internationally conducted research and meta analyses, prevalence of pre-pubertal bipolar disorder in those that develop clear cut bipolar is between 1.8 and 2.8 percent. More first episodes begin to emerge in adolescence.
- Cyclothymia:
mild depressions and hypomania (note: this less severe version of bipolar can
become worse with time and up to one-half of people with cyclothymia eventually convert to
Bipolar I or Bipolar II).
A complication of
bipolar disorder affecting about 20% of suffers is called rapid cycling.
This represents a time limited worsening of the illness in which episodes occur
with greater frequency (i.e. 4 or more episodes of depression, mania or
hypomania per year). Most cases of rapid cycling last a few months to a year
and a half and then subside. The most
common cause for rapid cycling is substance use/abuse or the use of antidepressants. If more frequent
episodes are evident it is referred to as ultra-rapid cycling or ultradian
cycling bipolar.
Untreated or poorly
treated bipolar illness leads to disaster. Careers and marriages are ruined,
physical health problems abound, and there is a high rate of suicide (19-20%
lifetime risk). If not treated, most cases of bipolar disorder become
progressively worse, likely owing to kindling effects. The sooner this illness
can be diagnosed and properly treated, the better.
Treatments for
Bipolar Disorder
Although the focus
of this course is on psychopharmacology, we will also briefly address
adjunctive treatments. Medication treatment alone is never adequate to fully
control bipolar disorders. Treatment must have a two-pronged focus: bringing to
an end the current manic or depressive episode and relapse prevention. With
proper medical treatment, most people can experience a marked decrease in
episode frequency and severity.
Lifestyle Management
People with bipolar
illness have a very unstable and fragile neurobiologic mechanisms for affect
regulation and extreme emotional lability and mood episodes can be triggered by
a number of environmental, psychological and physiological stressors. Before a discussion of medication treatment,
we will address lifestyle management issues. It is especially important to
regulate one's lifestyle closely: without this, medical treatments often are
only partially effective (Malkoff-Schwartz, et al. 1998). Most important
are:
- Maintain regular bed times and awakening
times. Such regularities in sleep patterns are crucial
- Avoid substance abuse and alcohol
use/abuse like the plague (substance abuse is very common in bipolar
disorder and often significantly aggravates the illness)
- Avoid sleep deprivation, shift work and
crossing time zones
- Avoid or greatly minimize caffeine use
since it can significantly disrupt the quality of sleep
- Keep the amount of bright light exposure
(e.g. sunlight) and the amount of physical exercise consistent each day year-round.
Medication Treatments: General Considerations
General references:
- Hirschfeld, et al. (2002): Bipolar disorder Practice Guidelines
- Goodwin and Jamison Manic Depressive Illness (2007)
- Texas Department of Mental Health (1998) Texas Medication Algorithm Project
Obviously, these official practice guidelines listed above are clearly dated. Newer guidelines are in development. Listed below are guidelines drawn from the list above but also include guidelines recommended by individual leaders in the field. The choice of medications used to treat bipolar disorder depends on the mood state the patient is currently experiencing (i.e. whether it is mania or depression). In addition, the medication choice always must take into consideration the ultimate goal of preventing recurrences and a lot of this ultimately depends on long-term medication tolerability.
Currently there are fourteen medications that are approved by the Food and Drug Administration (FDA) for the treatment of bipolar disorder: lithium, Thorazine, Depakote, Lamictal, Symbyax, Risperdal, Seroquel, Equatro, Geodon, Abilify, Saphris, Latuda, Rexulti, Vraylar, and Zyprexa. However, a number of other highly effective drugs are in common
use. The use of medications not approved by FDA for the treatment of certain
conditions is referred to as “off label use”…and it must be emphasized that
off label use of medications is very common practice in every branch of medicine (e.g. Haldol has been used to treat mania for decades, although it has never gotten FDA approval for treating this disorder).
Recent surveys reveal that in the United States only 11% of people being successfully treated for bipolar disorder are taking just a single drug (i.e. mono-therapy);
thus, this is the exception and not the rule. On average, most people being
treated are taking 3 or 4 medications simultaneously. The reason for this is
simple: medication combinations are often clearly superior to mono-therapy for
most people suffering from bipolar disorder.
All medications have
side effects and unfortunately the drugs used to treat bipolar disorder are
known to produce significant side effects for the majority of people being
treated. Side effects, at times, are mild and easy to tolerate. But often they
are more noticeable and in rare instances they can be dangerous. In every
single case, once the current mood episode has subsided, people with bipolar
disorder must continue to take certain medications (mood stabilizers) to help
prevent or reduce the likelihood of recurrence. This is absolutely essential!
However, some estimates suggest that as many as 90% of people who start medical
treatment for bipolar disorder will recover from their first episode, but
within weeks or several months, will stop taking the medications (against
medical advice). The most common reasons for doing so are understandable: 1.
patients are plagued by unpleasant side effects; 2. there is the negative stigma regarding mental illness; and/or 3. they conclude that
the episode they experienced is not really bipolar disorder, but was just a
single episode and that there will not be recurrences. This conclusion is borne
of hopefulness that this is not really going to be a recurring illness (Pope
and Scott, 2003). These reasons for discontinuing the medication are entirely
understandable, but they almost invariably lead to the emergence of another
episode (this may occur within a few months following the initial episode, but
more commonly occurs several years later).
For many patients, taking medications when you feel well, is counter-intuitive.
However, the picture is clear that bipolar disorder is always recurring, and
over a period of time there is a tendency for episodes to become increasingly
severe and harder to treat. There is also research that reveals that untreated
or poorly treated bipolar illness can ultimately result in lasting damage to
the nervous system. During mood episodes there are often toxic levels of certain
neurotransmitters (e.g. glutamate), increases in intracellular calcium and
stress hormones (e.g. cortisol) that are released, all of which can damage nerve cells.
Fortunately, studies also reveal that ongoing treatment with some bipolar medications
may prevent this from happening (Dreven, et al. 2002). In a very real sense,
some of these drugs (e.g. lithium, Depakote, Tegretol and Equatro) appear to be “neuroprotective”.
Many side effects
can be managed by dosage adjustments or by switching to other medications. This
is one reason that most times people will need to go through systematic trials
on a variety of medications to determine which ones are the most effective and
also which drugs are best tolerated for any given individual. Every effort
should be made to find the right medication or medication combinations in an
attempt to minimize side effects. And this is often something that can be
accomplished. However, it is often the case that it takes a year of trials on
various medications to finally discover the specific medication or medication
combinations that will be effective and that will be best tolerated. This is
the rule and not the exception…it is very important for patients to not feel
too discouraged if the first medications used are less than optimally effective
or if they have problematic side effects. A sign of a competent and
compassionate psychiatrist is his or her willingness to be persistent in
carrying out systematic medication trials until the best treatment is finally
identified. Sometimes side effects can be minimized, however, many people end
up having to find ways to tolerate some side effects. Obviously, this is not
pleasant, but is ultimately necessary to reduce or eliminate severe mood
swings. And unfortunately, a very small number of people are simply unable to
tolerate any bipolar medications.
Medication Treatments: What are Realistic Outcomes?
Bipolar disorder is like a number of other chronic medical conditions (such as emphysema, asthma, arthritis, etc.). It is not a condition that can be cured by currently available medications. However, the medications discussed below can be effective in relieving many of the more serious symptoms of bipolar illness and often can reduce the frequency of mood episodes for most people, if patients receive appropriate treatment and stick with it. Good news and not so good news: with aggressive, appropriate, and ongoing medication treatment, and if the treatment is started during the first or second mood episode, about 25% of people will not experience major recurrences. That is, in about one out of four people the medications are successful in preventing relapses (please note: if the first appropriate medical treatments begin after the second episode, typically treatment becomes somewhat more challenging and the outcomes are not quite as robust). However, for the majority of other people receiving treatment after the first episode, if they stay on medications the recurrence rates for severe episodes can be reduced by about 75% and hospitalizations can often be avoided. Subsequent episodes that do occur tend to be mild depressions and hypomanias (Gitlin, 2002). However, the majority of people being treated for bipolar disorders are frequently non-adherent with medical treatment and recurrent episodes are the rule, not the exception. One of the keys to long-term treatment success rests on medication tolerability. This, however, is a significant challenge since most bipolar medications carry high risks of unpleasant side effects.
Medication
treatments are far from perfect, but it is the kind of effectiveness that can
substantially reduce suffering, keep families together, avoid catastrophes and
save lives.
Bipolar Medications
There are six major classes of psychiatric medications that have been found
to be effective in treating various symptoms of bipolar disorder. Generic and
brand names (registered trademarks) and typical adult daily doses are listed
below.
Lithium:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
lithium |
Lithonate, Eskalith |
600-2400 mg |
Therapeutic blood levels
| Acute
mania: |
0.8-1.2 mEq/l |
| Prophylaxis: |
0.6-0.8 mEq/l |
Anticonvulsants mood stabilizers:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
divalproex |
Depakote |
750-1500 mg |
| carbamazepine |
Tegretol; Equatro |
600-1600 mg |
| oxcarbazepine |
Trileptal |
1200-2400 mg |
| lamotrigine |
Lamictal |
50-200 mg |
Therapeutic blood levels
| Depakote blood levels: |
50-125 mcg/ml |
| Tegretol; Equatro blood levels: |
4-12 mcg/ml |
| Trileptal blood levels: |
not yet established |
| Lamictal blood levels: |
not necessary to monitor |
Second Generation Antipsychotics (SGA): also referred to as atypical antipsychotics
(the name commonly used for a class of antipsychotic medications that have been developed since the early 1990s that treat psychotic symptoms and also have anti manic effects as well; two SGAs: Latuda and Seroquel, also treat bipolar depression).
Generic Name |
Brand Name |
Typical Adult Daily Dose |
olanzapine |
Zyprexa |
5-20 mg |
| risperidone |
Risperdal |
4-10 mg |
| ziprasidone |
Geodon |
60-160 mg |
| aripiprazole |
Abilify |
15-30 mg |
| quetiapine |
Seroquel |
150-400 mg |
| paliperidone |
Invega |
3-12 mg |
| iloperidone |
Fanapt |
12-24 mg |
| asenapine |
Saphris |
10-20 mg |
| lurasidone |
Latuda |
40-80 mg |
| brexpiprazole |
Rexulti |
1-4 mg |
| cariprazine |
Vraylar |
1.5-6 mg |
Antidepressants/Antipsychotic combinations
Note: Symbyax, a combination of Prozac and Zyprexa,
was approved by the FDA in 2004 for the treatment of bipolar depression. It
comes in the following formulations: (Zyprexa dose/Prozac dose): 6/25 mg., 6/50
mg., 12/25 mg., 12/50 mg.
Calcium Channel Blockers
Generic Name |
Brand Name |
Typical Adult Daily Dose |
verapamil |
Calan, Isoptin |
360-480 mg |
Benzodiazepines
(also referred to as
minor tranquilizers or anti-anxiety drugs; only listed are those in most common
use)
Generic Name |
Brand Name |
Typical Adult Daily Dose |
diazepam |
Valium |
4-30 mg |
| clonazepam |
Klonopin |
0.5-2 mg |
| lorazepam |
Ativan |
2-6 mg |
Note: Alproazolam (Xanax) is the one tranquilizer that can aggravate mania.
Benzodiazepine
and benzodiazepine-like Sleeping Pills:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
temazepam |
Restoril |
15-30 mg |
| triazolam |
Halcion |
0.25-0.5 mg |
| zolpidem |
Ambien |
5-10 mg |
| zaleplon |
Sonata |
5-10 mg |
| eszopiclone |
Lunesta |
1-3 mg |
Non-benzodiazepine sleeping pills:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
ramelteon |
Rozerem |
4-16 mg |
| suvorexant |
Belsomra |
15-40 mg |
Note: In the past two years the National Sleep Foundation and the National Institute of Health have issues warnings that high dose melatonin (i.e. 2 mg and higher), may destabilize both depression and bipolar disorder.
Targets for Medication Treatment
There are three
primary goals in medication treatment of bipolar disorder: dealing with
potentially dangerous emergency issues (e.g. extremely severe agitation or
suicidal impulses), resolving the current episode (whether mania or
depression), and relapse prevention. The choice of medications used always will
be influenced by these goals. In addition, and obviously the medication choice
will also be dictated by the need to minimize side effects.
Getting Started with Medication Treatment:
Emergency Medication Treatments and Laboratory Tests
Sometimes there is a
need for emergency treatment; for example, if a person is experiencing a sudden
onset of severe manic agitation (which may include extreme restlessness,
impulsivity, severely impaired judgment and/or aggression) or serious suicidal
impulses during a depression. At such times acute medical treatment may be
necessary.
When there is such a
crisis, hospitalization is almost always necessary. Emergency medical
treatments for agitation include the use of either benzodiazepines
(anti-anxiety tranquilizers, such as Ativan, Klonopin, and Valium) or
antipsychotic medications (such as Zyprexa, Risperdal, or Haldol). These two
classes of drugs are often very effective in rapidly reducing agitation. On
occasion there is a need for emergency medical treatment for very severe
depression (where there is either a grave suicide risk or refusal to eat
accompanied by severe weight loss). In such cases ECT (electro-convulsive
therapy; “shock” treatments) can be successfully used. ECT is also very
effective for the emergency treatment of severe mania. The use of ECT in treating severe mania is very limited.
If the situation is not extremely urgent, then it is commonplace to order some
pretreatment laboratory tests. This is done for two purposes. The first is to
rule out the possibility that the mood symptoms may be caused by a primary medical
illness (such as thyroid disease) or substance abuse (e.g. to see if manic symptoms are being caused by methamphetamine use). The other reason has to do with the tendency
for many of the bipolar medications to cause significant changes in a variety
of bodily functions. Mood stabilizers in particular are known to affect a broad
range of organs and glands especially when they are taken for prolonged periods
of time. Thus typically, pretreatment labs include measures of cardiac, kidney,
liver and thyroid functioning as well as a complete blood count. Laboratory
monitoring of blood levels of certain medications may also be required. This
is routinely done for the following mood stabilizing medications: lithium, Tegretol,
Trileptal, Equatro and Depakote.
Treatment Guidelines: Mania
Several classes of
psychiatric medications have been found to be effective in treating acute manic
episodes:
- Lithium
- Anticonvulsants: Depakote, Tegretol, Equatro, Trileptal
- Antipsychotic medications: Second generation antipsychotics (SGAs: see chart above) and Haldol (this final drug is an older generation antipsychotic that is still sometimes
used)
- Calcium Channel
Blockers: Verapamil (generally only used for treating mania in pregnant women
because it is the safest mood stabilizer for use during pregnancy)
- Benzodiazepines
(minor tranquilizers):
Benzodiazepines and
antipsychotic medications are given initially to reduce agitation, which can
often be achieved within a few hours (note: for reasons that are not well
understood, as noted above, the tranquilizer, Xanax can sometimes aggravate mania, and thus should not be used to treat acute mania). The anti-manic medications (e.g.
lithium, SGAs, and anticonvulsants) generally require 7-10 days of treatment before you see an
onset of action and symptom reduction. Once symptoms begin to be reduced,
continued treatment for several weeks will often be necessary to eliminate
acute manic symptoms. As noted earlier, most people will ultimately be treated
with 3 or 4 medications simultaneously to achieve the best outcomes.
There are three
stages in the medical treatment of mania:
- Reduce extreme
agitation (the goal is to get agitation under control within a few hours).
Severe agitation can be dangerous to the patient as well as to others around
them and this must be addressed as soon as possible.
- Reduce core
manic symptoms such as restlessness, sleeplessness, rapid speech, paranoid
ideas, etc.
- Begin treatment
for relapse prevention.
Stage One
As mentioned above, antipsychotic medications and benzodiazepines are the best medications for treating acute agitation; they quickly produce substantial sedation, calming, or sleep. It is important to note that although antipsychotic medications do successfully treat psychotic symptoms (such as hallucinations) they also are effective anti-manic agents. Most mood stabilizers require the 7-10 day period of time before symptom reduction, but with one notable exception: the anticonvulsant mood stabilizer, Depakote, when given in large doses, can begin to show anti-manic effects in about 4 days. Once severe agitation has subsided, often benzodiazepines are gradually reduced and then within a few days are discontinued. This may also be true for antipsychotic medications. However, often antipsychotic drugs may continue to be used for a prolonged period of time.
During stage one of
treatment, as mentioned above, a number of lab tests are often done to monitor
the early effects of the drugs.
Stage Two
The choice of
medications used to treat core symptoms of mania is important and often
complex. As noted above, there are several different types of mania and a
considerable amount of research has been done to discover which medications are
best suited for treating particular subtypes of mania. Dozens of large-scale
research studies have been conducted in recent years and specific treatment
guidelines have been developed that are very useful in helping physicians to
decide on initial medication choices (see below). However, the fact is that
each person will have a number of factors unique to her or him that will
influence the choice of medications, such as age, gender, body weight, history
of allergies to medications, liver metabolism rate, the presence or absence of
other medical conditions and other medicines being used to treat such
conditions. Your patients must anticipate that it is extremely common for
psychiatrists to make initial medication choices, begin treatment and then
during the following weeks or months make what are often frequent changes in
the doses or medications prescribed. There is an important reason for
emphasizing this. Many times, people being treated for bipolar illness or their
family members become worried as they begin to encounter side effects, or they
must go through what seems like an endless number of lab tests or changes in
medications or medication doses. Many people become concerned that these
medication changes suggest that their doctor may not be competent or that their
case of bipolar is especially treatment-resistant. This then can lead to
discouragement and feelings of pessimism. Here is the truth: the pathway to
recovery and good outcomes, more often than not, is complicated. The rule, not
the exception is that people will be tried on several if not many medications
in the search for the right drug or medication combinations. It is so important
to help patients understand this and not conclude that the frequent changes in
medications are necessarily a reason for concern. The fact is that bipolar
disorder is challenging to treat and often requires a considerable amount of
time systematically trying various medications before the right medications
combinations are found.
“Classic Mania” (with euphoria, expansiveness, upbeat mood, irritability, etc.)
has been found to respond best to treatment with lithium or Depakote (other
anticonvulsant mood stabilizers or atypical antipsychotic medications often
can treat classic mania, but in head-to-head comparisons, lithium and Depakote
appear to be the best first-line medications for this type of mania). Generally
during stage two of treatment, especially if this is a person’s first episode
of mania, just one of these medications will be prescribed. Assuming that the
medication is tolerated (i.e. that side effects are mild or manageable) treatment
will continue for a period of several weeks. As mentioned earlier, 90% of people
being treated for bipolar disorder ultimately must take two or more medications
at the same time to adequately treat mania. Thus, it is possible that the one
medicine initially prescribed may be tolerated and may eventually be effective.
Decisions to increase the dose or to change or add another medication in the
ensuing weeks will depend on tolerability and effectiveness. A trend in treatment is to begin with two anti-manic medications relatively early in treatment (since most times this is ultimately what is required to achieve success) and by administering both drugs (e.g. Depakote and lithium) each at somewhat lower doses, the two medications together often produce fewer side effects than a single agent used at high doses. Since there are
always possible drug-drug interactions, then generally the recommended approach
is to first optimize treatment with one medication (which means to progressively
increase the dose while always being watchful for the emergence of side effects).
What is hoped for is that the first signs of symptomatic change will occur during
the first 7-10 days and that symptomatic improvement will continue to unfold
over the next few weeks. Just how long it takes to fully resolve a manic episode
varies from one individual to another.
Should side effects
be significant, typically there will be either a dosage adjustment or possibly
a change to another medication. If side effects are mild to moderate and
tolerable, but there is only partial improvement in symptoms after several
weeks of treatment, then a decision will be made to either change to a
different medication or to add another medication (i.e., augmentation). Medications typically used for
augmentation include anticonvulsant mood stabilizers and/or antipsychotic
medications.
Mania with Mixed Features: (agitation, decreased need for sleep, extreme irritability, insomnia, rapid speech, feelings of despair, hopelessness, etc.). There is some controversy regarding the treatment of dysphoric mania. However, most experts agree that the best first-line medication is Depakote. Many people experiencing dysphoric mania do have positive responses to lithium as a mono-therapy. The use of just one medication again, initially is the typical strategy and again, before adding or changing medications, the drug used will be optimized. As in the treatment of classic mania, we are looking for the first signs of improvement within the first 7-10 days.
If after several weeks of treatment and if increased doses of the medication
yield only partial symptomatic improvement then augmentation can be used. Often
the first augmentation strategy is to combine Depakote and either lithium or an antipsychotic. If other
medications are required then the addition of the following are commonly prescribed:
anticonvulsants: Tegretol, Equatro or Trileptal. The anticonvulsant,
Neurontin has been found to be ineffective as a mono-therapy, but it is often
used as an augmenting agent, especially helpful in reducing anxiety (50% of
bipolar patients have comorbid anxiety disorders). Other choices include atypical
antipsychotics.
The treatment of childhood-onset bipolar disorder is beyond the scope of this
CE course; however, a few brief comments will be made. When mania occurs in pre-pubertal
children, it almost always presents as a form of dysphoric mania with rapid
cycling and marked irritability. With adults the general strategy is to begin
treatment with one mood stabilizer and only later add additional medicines if
they are needed (although as noted above sometimes two drugs are started very early in treatment). Preliminary research has rather strongly indicated that most children suffering
from mania ultimately end up taking two or more mood stabilizers (this is required
for most to effectively eliminate manic symptoms). Thus, there currently is a trend to begin treatment with children using two mood stabilizers (often this combination is Depakote and lithium). It is generally felt that
the much higher success rate with two mood stabilizers outweighs the added side
effects of using two drugs. Also, it is felt that the earlier you can put a
lid on mania and arrest its development, the better…to do so matters not only
regarding the current episode but may also have a positive effect on reducing
the severity of future episodes.
Rapid Cycling
As mentioned above,
rapid cycling generally is a period of time lasting anywhere from a few weeks
to a year or year and a half where there is a significant increase in the
frequency (and often) severity of mood episodes. In only about 2% of people is
rapid cycling continuous for very prolonged periods of time. Three factors
account for the majority of cases of rapid cycling: substance abuse (including
alcohol), the use of certain prescription medications (e.g. antidepressants,
steroids, stimulants), or disorders of the thyroid gland. Thus, it is very
important to determine whether or not any of these factors are present and take
appropriate action to ameliorate them. Beyond this, special attention must be
taken to stabilize the patient’s lifestyle, especially making sure that there
is regularity to one’s sleep patterns and making every attempt to reduce or
avoid sleep deprivation (e.g. establishing regular bed and awakening times,
completely avoiding sleep destroying substances such as caffeine, alcohol and
decongestants, no pulling all-nighters cramming for exams or late-night
partying).
Beyond these strategies, the following medication strategies have been found to be helpful. Preferred mood stabilizers include: Depakote, Tegretol, and Lamictal. Atypical antipsychotic medications (SGAs) are frequently also prescribed. Finally, the most common mood symptoms seen in rapid cycling are depression or a combination of depression and irritability. However, antidepressants can, unfortunately, contribute to cycle acceleration and rapid switches in mood. Thus the use of antidepressants in rapid cycling is not indicated.
Treatment-Resistant
Mania
For people who
experience very severe mania that does not respond to more traditional
treatments there are a number of options.
The antipsychotic
medication clozapine (brand name Clozaril) has been found to be effective in
some cases of treatment resistant mania. This drug has antipsychotic effects
(e.g. for treating hallucinations, delusions, etc.). It is proving to be
effective for treating not only mania but
relapse prevention. Unfortunately, Clozaril is plagued by numerous, significant
side effects, some of which are potentially dangerous. ECT (electro-convulsive
therapy) is a safe and highly effective treatment for severe mania.
Stage Three: Relapse Prevention
Once the current
manic episode is completely controlled it is common practice to continue
medications, even though there are no obvious symptoms. This is necessary
because it is clear that once symptoms subside, if one discontinues then, the
acute relapse rate can be as high as 85%. Thus for a period of several months,
typically, medication treatment is continued and often at the same doses used
during treatment of the acute phase of the episode. This phase of treatment
appropriately is called continuation treatment. After several months, assuming
there have been no “breakthrough” symptoms, then the next stage of treatment,
maintenance treatment begins. Here the focus of treatment is on the prevention
of recurrent episodes. Often if a person has been receiving lithium, the dose
is gradually reduced (which often results in fewer side effects). The doses of
other medications may also be reduced slightly, however such a decision is highly
individual and is influenced by a number of factors including a person’s
clinical history and the presence of particular side effects.
In general, the medications used to treat mania are considered to be very effective
for most people experiencing a manic episode (Note: this is true for bipolar
mania seen in patients who have a late adolescent or adult-onset illness. Mania
in pre-pubertal children is significantly more difficult to treat). However,
the more long-term goal of preventing recurrences is more challenging. Despite
the fact that there have been decades of experience in treating bipolar illness,
there are no good long-term studies on relapse prevention (the longest studies
available only extend to about a year). Yet this is a lifelong illness and all
experts agree that lifelong treatment is required. It is important to know that
most medications used to treat bipolar disorder do have side effects that may
emerge with very long-term use, thus necessitating periodic lab tests to monitor
blood and various glands and organ system functioning. What is clear is that
failure to treat (or to adequately treat) bipolar disorder almost always leads
to disasters.
The medications for which the best data exist for long-term maintenance treatment
are the following mood-stabilizers: lithium (the best data), Lamictal and
Depakote. Most people on maintenance treatment will continue to
take several medications. However, longer-term treatments generally do not include
treatment with benzodiazepines or antidepressants. These medications may destabilize
bipolar patients. In the end what matters is the ability to find medication combinations that achieve long-term tolerability.
Treatment Guidelines: Bipolar Depression
Several classes of
psychiatric medications are often used to treat bipolar depression:
- Lithium (to treat bipolar depression lithium levels generally have to reach 0.8 or higher)
- Symbyax (a combination of Zyprexa and
Prozac…FDA approved to treat bipolar depression)
- Lamictal (note: among bipolar medications, Lamictal has the most favorable side effect profile)
Atypical
antipsychotic medications may have antidepressant properties:
- In particular, Abilify, Latuda and Seroquel have antidepressant effects in bipolar depression.
It is important to underscore that many treatments that ordinarily are effective in
reducing unipolar depression (e.g., antidepressants) carry a risk of provoking
manic episodes (a phenomenon referred to as switching) or causing cycle acceleration
(this refers to a gradual, over-all worsening of bipolar disorder in which,
over time, there is an increased frequency of episodes and episodes tend to
become more severe and more difficult to treat). Switching and cycle acceleration
have been documented with the use of antidepressants
and thus these drugs should only rarely be used in the treatment of bipolar depression
(Ghoemi, et al. 2001; Post, et al. 2001). Some recent studies suggest that the addition of an antidepressant to a mood stabilizer may be effective in treating treatment-resistant bipolar II patients, but this approach is not recommended in the treatment of Bipolar I. Excessive bright light exposure (which
can treat some types of seasonal depression) has also been associated provoking
manias. Additionally, three popular over-the-counter products that have antidepressant
properties may, likewise, cause switching or cycle acceleration: St. John’s
Wort, 5-HTP, and SAM-e.
The general strategy in treating bipolar depression is to first “do no harm”…do
not use antidepressants. We will see, however, there are some exceptions.
There are also three stages in the treatment of bipolar depression.
Stage One
In the event of life threatening symptoms
such as strong suicidal impulses or refusal to eat, ECT is a highly effective
treatment. The treatment approach is much the same as used to treat acute
mania. The other emergency treatment is hospitalization. Unfortunately, aside
from ECT, most approaches to treating depression require several weeks before
one is likely to see symptomatic improvement.
Stage Two
Start with a medication with proven efficacy in treating bipolar depression.
The drugs that have the best track record of effectiveness, as noted above, are Seroquel, lithium, Symbyax and Lamictal (note: Latuda was approved to treat bipolar depression in late 2013 and it remains to be seen how effective it is after a number of months being used in clinical practice). Of these medications, Seroquel is likely
to be the most effective. Lamictal is often also effective in treating bipolar depression (however, there is an important consideration when
using Lamictal: as treatment is begun, it is required that Lamictal be given
in small doses and that dosage changes are done very gradually during the first
4-6 weeks of treatment. This is done to avoid inducing a potentially dangerous
rash [Stevens-Johnson syndrome] that can occur if there is rapid dose escalation; Fortunately, since the use of gradual dosing has been adopted, the risk of Stevens-Johnson is almost nil; this is the case with older teenagers and adults; Stevens-Johnson can still occur in pre-adolescent children and should generally not be used in this population). Lithium has been shown to have antidepressant effects when administered
as a monotherapy in bipolar depression (although not in unipolar depression);
however, as noted above, the blood level of lithium that is effective in combating depression appears to require at least a 0.8 level. Just how urgent the need is for antidepressant action, will be a critical factor in choosing Lamictal, Latuda, Seroquel, lithium or Symbyax (i.e. due to the need to slowly increase the dose of Lamictal, it may take somewhat longer to have an effect where with lithium, Seroquel, Latuda, or Symbyax doses can be more rapidly titrated up). If there is any history of rapid cycling, switching or a recent manic episode, it is best to start with one of the following medications since each also have anti-manic properties: lithium, Latuda or Seroquel. Although Symbyax contains an antimanic agent (Zyprexa) it also includes an antidepressant (Prozac), which may provoke switching). There have been a few case reports of Lamictal inducing mania.
If treatment with the first-line medication is not successful or there is a
partial response then there are several options:
- Combine two of these medications
- ECT
Stage Three
This is much the same as stage three treatments for mania. The bipolar medications that have the best record of effectiveness in the prevention of recurrent depressive episodes are
Lamictal, Seroquel and lithium.
Adjunctive Therapies
Bright light therapy
(using a commercially available light box which generates 10,000 lux of light
intensity for 10-30 minutes a day) has been used for treating bipolar
depression, especially for those who routinely have winter depressions or who
work the night shift. This treatment is typically combined with medication
treatments and like all treatments for depression; it too carries a risk of
provoking mania in people with bipolar disorder.
Omega-3 Fatty Acids: Approximately 60% of the human brain is composed of lipids (fats) and between 30-35% of brain mass is made up of omega-3 fatty acids. These molecules are important in forming cell membranes, synapses and in facilitating nerve cell actions. The most abundant dietary source of omega-3 fatty acids is fish and shellfish. In cross-cultural studies it has been found that in countries where people eat a lot of fish and other seafood, the severity of mood disorders is less. This interesting finding led researchers to carefully evaluate the impact of diet on mood. During the past ten years a number of studies have been conducted with people suffering from bipolar disorder and also major depression. Unfortunately, omega-3 fatty acids have not been shown to be effective as an augmenter to standard mood stabilizers. However, people with bipolar disorder suffer from high rates of heart disease and strokes, and thus omega-3, which can afford some protection against vascular disease, may be helpful in that respect.
Exercise therapy has
recently been shown to be effective in treating major unipolar depression, however, to
date there are no studies of this approach in treating bipolar depression.
High intensity light therapy (formerly used only to treat seasonal depressions) has been shown to be effective in treating all depressions. Caution: excessive exposure to high intensity light may provoke mania in depressed people who suffer from bipolar disorder.
Psychotherapy
Although medication treatment is the backbone of successful therapy for bipolar, a number of studies have clearly shown that psychotherapy (especially Interpersonal and social rhythm therapy, Cognitive-behavioral therapy or Family-based Psychoeducational therapy) can significantly contribute to better treated outcomes.
Anxiety Disorders
General
Considerations
Anxiety disorders affect 25% of people (lifetime prevalence)
and many become very chronic conditions. All anxiety disorders discussed in
this section have been found to respond well to exposure-based cognitive
therapy. In most instances this form of psychotherapy is more effective than
drug treatments. Despite this, medication treatments are often useful or
necessary owing to the following:
- In many
communities there may not be professionals trained to provide cognitive therapy
- Some patients
refuse cognitive therapy owing to the fact that such treatments require
exposure, and people are too afraid to undertake the treatments.
- Often, symptoms
are so debilitating that medication intervention is an important first step to
take to reduce suffering and help people re-engage in normal life functioning
and then begin psychological treatments.
- Some patients
with marginal ego strength may not be able to tolerate exposure-based
treatments.
- If medications
are used and at some point, exposure-based treatments are employed, it will be
necessary to gradually reduce the dose of medications so that the client will
experience some anxiety. This is required since it has been found that
exposure therapy and deconditioning is not successful unless the person
experiences some amount of anxiety, is able to undergo exposure trials using
anxiety management skills, and have the experience of enduring anxiety without
catastrophe. This is essential in order to experience mastery.
Diagnostic Issues
The following seven classes of anxiety disorders will be
discussed:
- Generalized
Anxiety Disorder: chronic, pervasive, low-level anxiety characterized by
excessive worry.
- Stress-related
Anxiety (i.e. adjustment disorder with anxiety): anxiety symptoms have only
emerged in the wake of a recent, significant life stressor. Listed in DSM-5 under the chapter: trauma-related disorders.
- Panic Disorder:
Characterized by repeated episodes of panic that erupt and rapidly escalate
into intense anxiety within 10 minutes or less and then dissipate. Often
accompanied by anticipatory anxiety and phobic avoidance (e.g. agoraphobia).
- Social Anxiety:
anxiety symptoms are only experienced in social settings, e.g. social
gatherings, public speaking, etc.
- Obsessive-Compulsive
Disorder: presenting with recurrent, debilitating obsessions and compulsive
rituals. Note: OCD is discussed in a separate chapter in DSM-5.
- Acute Stress
Disorder (ASD) and Post Traumatic Stress Disorder (PTSD): ASD becomes PTSD if
symptoms persist beyond one month. Symptoms include: intrusive symptoms
(memories, flashbacks, nightmares, intense emotional responses/reactivity),
avoidance, numbing and dissociation, persistent hyperarousal, and often
depression. Occasionally transient psychotic symptoms may be present. Note: ASD and PTSD are discussed in a separate chapter in DSM-5: trauma related disorders.
- Anxiety due to
a general medical condition or substance use or abuse. Of note here is that the
use of caffeine can cause or significantly exacerbate anxiety symptoms. In
every case presenting with anxiety, it is critical to evaluate the presence of
caffeine use. The following questionnaire can be helpful in screening for
caffeine use.
Click here for the printable Caffeine Questionnaire
CAFFEINE CONSUMPTION
QUESTIONNAIRE |
| |
|
|
Average number of servings/doses/tablets
per day |
|
Average total per day |
| Beverages |
| Coffee (6 oz.) |
125 mg |
X |
|
= |
|
| Espresso (one oz.) |
50 mg |
X |
|
= |
|
| Decaf Coffee (6 oz.) |
5 mg |
X |
|
= |
|
| Tea (6 oz.) Black |
50 mg |
X |
|
= |
|
| Green Tea (6 oz.) |
35 mg |
X |
|
= |
|
| Energy Drinks |
200 mg (equivalent*) |
|
|
|
|
| Cocoa (6 oz.) |
15 mg |
X |
|
= |
|
| Caffeinated Soft Drinks (12 oz.) |
40-60 mg |
X |
|
= |
|
| Chocolate Candy Bar |
20 mg |
X |
|
= |
|
| Over-the-Counter Medications |
| Anacin |
32 mg |
X |
|
= |
|
| Appetite-Control Pills |
100-200 mg |
X |
|
= |
|
| Dristan |
16 mg |
X |
|
= |
|
| Excedrine |
65 mg |
X |
|
= |
|
| Midol |
132 mg |
X |
|
= |
|
| NoDoz |
200 mg |
X |
|
= |
|
| Triaminicin |
30 mg |
X |
|
= |
|
| Vanquish |
33 mg |
X |
|
= |
|
| Vivarin |
200 mg |
X |
|
= |
|
| Prescription Medications |
| Cafergot |
100 mg |
X |
|
= |
|
| Fiorinal |
40 mg |
X |
|
= |
|
| TOTAL MG. CAFFEINE PER DAY |
|
*Caffeine content of energy drinks vary. They also include a number of stimulating herbs.
> 250 milligrams a day, if taken after noon, may interfere with deep sleep
If caffeine use exceeds 250 mg. per day, there is a likelihood that it will
interfere with slow wave/deep sleep and may play a role in increasing any psychiatric
symptom. When anxiety is a prominent feature, then caffeine use should be completely
avoided. Many individuals that suffer from anxiety disorders inadvertently consume
significant amounts of caffeine and this always complicates psychological or
medical treatment. “Energy drinks” should also be avoided.
Psychopharmacology
There are two major classes of medications used to treat
anxiety disorders: antidepressants (all mentioned above with the exception of
Wellbutrin, which can increase anxiety) and the minor tranquilizers (commonly
referred to by their class name: benzodiazepines). We will also consider
adjunctive and experimental treatments.
Antidepressants have been addressed in detail above, thus there will be only
a few issues presented here. Most of the antidepressants (especially those that
target serotonin) have been found to be highly effective in treating most forms
of anxiety disorder. However, all have the tendency to produce initial activation (affecting 10-20% of people starting these medications).
This is a side effect of the drugs that can appear within a few hours after
taking the first dose. This is very problematic for those with anxiety disorders.
The increase in anxiety that occurs due to activation ranges from mild to marked
and typically lasts for 10 days, and then begins to subside (although in some cases it may persist for a longer period of time). Most patients who
encounter this become frightened by the increase in anxiety and stop taking
the antidepressant. Thus, a popular and effective way to address this is to
co-administer a benzodiazepine. The tranquilizer, if dosed appropriately, begins
to reduce anxiety within 30-45 minutes and can also operate to override activation.
The goal is to use the benzodiazepine for the first month of treatment and then
to phase it out. After one month of treatment, most antidepressants begin to
show antianxiety effects. There is one note of caution. Since benzodiazepines
can be habit-forming, they should be used with great caution in patients who
have a history of alcohol or other substance abuse. In such individuals an alternative
to the use of a benzodiazepine is hydroxyzine (brand names: Atarax; Vistaril). This is an antihistamine that has significant antianxiety properties. Benadryl can also be used to treat initial insomnia.
Benzodiazepines
Benzodiazepines:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
diazepam |
Valium |
4-30 mg |
| clonazepam |
Klonopin |
0.5-2.0 mg |
| lorazepam |
Ativan |
2-6 mg |
| alprazolam |
Xanax |
1-4 mg |
Medications used to treat anxiety-related initial insomnia:
(See also, my course Bipolar Spectrum Disorders: Diagnosis and Pharmacologic Treatment.)
Generic Name |
Brand Name |
Typical Adult Nighttime Dose |
temazepam |
Restoril |
15-30 mg |
| triazolam |
Halcion |
0.25-0.5 mg |
| estazolam |
Prosom |
1-2 mg |
| zolpidem |
Ambien |
5-10 mg |
| zaleplon |
Sonata |
5-10 mg |
| eszopiclone |
Lunesta |
1-3 mg |
| diphenhydramine |
Benadryl |
25-50 mg |
| ramelteon |
Rozerem |
4-16 mg |
| survorexant |
Belsomra |
15-40 mg |
| doxepin |
Silenor |
3-6 mg |
Special Concerns: If
benzodiazepines are being taken on a regular basis the body develops a
tolerance for the medication. When this happens, typically the drugs continue
to work to reduce anxiety, but the problem is that when there is tolerance, if
one abruptly stops the medication there can be withdrawal symptoms. Withdrawal
symptoms usually include nervousness, agitation, difficulty falling to sleep,
and on occasion can produce seizures. This needs to be taken very seriously. If
a patient has been taking a benzodiazepine on a daily basis for more than 6
weeks and especially if the dose is moderate-to-high, withdrawal reactions are
a very real risk. One should never abruptly stop taking the medication without
first consulting with his or her physician. It is also a good idea to be
especially careful to monitor the supply of the medications so that refills can
be requested in a timely fashion. Many people find it helpful to keep at least
a two-day supply on hand in the event that it takes longer than usual for a
prescription to be refilled. Additionally, benzodiazepines can be abused and
should not be prescribed to people with a substance abuse history.
The onset of action
of benzodiazepines is 30-45 minutes. As noted above, most antidepressants
require a minimum of four weeks of treatment before the first signs of clinical
improvement are noted.
Generalized Anxiety Disorder
Three classes of medications have been found to be effective
in treating GAD: buspirone (BuSpar, a non-habit-forming tranquilizer),
benzodiazepines and antidepressants. BuSpar requires ongoing treatment for 2-6
weeks before the onset of clinical improvement and is given in doses of from
10-40 mg per day. The symptomatic improvement is seen to emerge gradually and
primarily is experienced by the patient as a decrease in worry and rumination.
BuSpar has a very favorable side effect profile and is well tolerated, however,
its efficacy is not as robust as that seen with benzodiazepines or
antidepressants. Benzodiazepines are effective in treating GAD, although there
has been some controversy regarding their use. Clearly, they are problematic for
three reasons: in patients with a substance abuse history they can be abused,
although this rarely occurs in the absence of such a history … (this warning will
apply to treatment of all anxiety disorders discussed below), they can cause
sedation and impairments in alertness, and finally, as noted above, there is
the potential for significant withdrawal effects should there be a rapid
discontinuation. Antidepressants (except for Wellbutrin) are generally very
effective in treating GAD but require 4-6 weeks of treatment, or longer in some
instances, before they begin to show clinical improvement (this is true for all
anxiety disorders discussed below, except for OCD which often requires a more
prolonged period of treatment before maximal symptomatic improvement is seen).
It is generally true for GAD and all of the other anxiety disorders, that when
drugs are eventually discontinued at least 50% of patients will have a
return of symptoms, and thus drug treatments in most cases target symptoms but
do not cure the underlying neurobiologic disorder. Doses used to treat GAD are
similar to those used to treat major depression (see above). Such dosing is the
case for all anxiety disorders discussed below with one exception: OCD often
requires very high doses.
An experimental treatment is consuming 6 ounces of chamomile tea once a day. There is no acute decrease in anxiety, but after several weeks of treatment, a gradual decreased level of generalized anxiety may be experienced. Subsequent studies show even more robust results using 1220-1500 mg at bed time (must be obtained in capsule form from health food stores).
Stress-Related Anxiety
Short-term use of low-to-moderate doses of benzodiazepines
can be helpful in reducing both anxiety and stress-related initial insomnia.
BuSpar and antidepressants are not indicated since they require a number of
weeks to begin working and most acute stressors subside more quickly.
Panic Disorder
Two classes of medications are effective in treating panic
disorder: antidepressants and benzodiazepines. The advantage to using
benzodiazepines is the quick onset of action. This can be a godsend for
patients plagued by frequent and severe panic attacks. The particular drugs
used most often are the high-potency benzodiazepines: Xanax, Ativan and
Klonopin (other benzos often cause too much sedation). Doses required to
contain panic attacks vary considerably and in every case the patient is
started on a low dose and gradually increased until symptom control is
achieved. It is necessary to have the medication in the body continuously, 24
hours per day to avoid panic attacks (i.e. they cannot be taken only on an “as
needed” or PRN basis since panic attacks come on suddenly and the drugs require
30-45 minutes to become active in the central nervous system). Antidepressants,
once they have begun to reduce anxiety, are the preferred treatment of choice
since they generally require only once a day dosing and usually do not create
sedation nor are they habit-forming. Once panic symptoms have been eliminated
or greatly reduced, behavioral treatment for phobias is always indicated (the
medications are very effective in reducing panic symptoms but not robust in
their impact on avoidance and phobias).
Social Anxiety
Social anxiety disorder can be treated with benzodiazepines
or antidepressants. The choice of treatments depends largely on how pervasive
the symptoms are. If they are only occasional and highly situation specific,
benzodiazepines may be preferred (additionally, in such cases the use of
Inderal, a beta blocker…taken one hour before exposure to a social situation at
doses of 10-40 mg. can reduce some autonomic symptoms of anxiety such as
trembling, rapid heart rate and perspiration. This medication is not indicated
for chronic use since it has a tendency to cause depression if used
continuously, but can be effective used on an “as needed” basis).
Antidepressants are effective in the treatment of more severe or pervasive
forms of social anxiety.
Obsessive Compulsive Disorder
OCD may respond to treatment with SSRIs or the tricyclic antidepressant Anafranil
(clomipramine. Daily doses: 150-250 mg.). As noted above, the treatment of OCD
often requires using antidepressant doses that are higher than that used to
treat depression (i.e. the high end of dosage ranges listed in the section on
depression, e.g. 60-80 mg of Prozac daily). And it has been found that ongoing
treatment will often yield gradual improvement during the first 12 months of
treatment. That is, initially reaching a high but tolerable dose and staying
with that dose for 12 months may continue to yield ever-increasing improvement
with a plateau being reached by 12 months. Anafranil has significant side effects
and thus is generally a second line treatment, although it has the most robust
efficacy of all antidepressants in the treatment of OCD. A common practice is
to start with an SSRI and if there is only partial improvement, augment with
low doses of Anafranil (e.g. 25-50 mg.). This strategy is often effective in
providing additional symptomatic improvement. Unfortunately, when medications
are discontinued for OCD 90%+ individuals experience a full recurrence of symptoms
and thus the medications are only suppressing symptoms.
Post-Traumatic Stress Disorder
Acute treatment of PTSD (or Acute Stress Disorder) is best accomplished by the use of clonidine or beta blockers which rapidly decrease anxiety symptoms. Traditionally benzodiazepines have been used, but several studies have actually shown that chronic treatment with tranquilizers can produce poor outcomes. Thus, use of benzodiazepines should be limited to very brief treatment (one or two days). PTSD has also been successfully treated with SSRIs. The down side of SSRI use is that it requires 3-4 weeks of treatment before symptom reduction is noticed and can, in some individuals cause initial activation (i.e. anxiety), as noted above, which can of course be very problematic. These medications reduce intrusive symptoms, hyperarousal and, interestingly, dissociation and numbing symptoms. As with OCD, sometimes relatively low doses may be effective, but more often the dose of antidepressants needed to provide symptomatic relief needs to be high. Case reports suggest that partial responders may benefit by adding BuSpar to the SSRI. Although anxiety can be pronounced with PTSD, generally benzodiazepines are not recommended for longer-term treatment and may in fact complicate recovery. If transient psychotic symptoms are evident, low doses of atypical antipsychotics may be helpful (e.g. 2 mg Risperdal or 7.5 mg. Zyprexa). Generally, the use of antipsychotics can be of relatively short
duration. Experimental drugs that may be effective in treating PTSD-related anxiety are beta blockers (e.g. Inderal), clonidine (Catapres; Kapvay), guanfacine (Tenex; Intuniv) and for treatment of nightmares, prazosin (Minipress). Prazosin is also being used to treat daytime anxiety in those suffering with PTSD.
Anxiety related to a general medical condition or substance use / abuse
In most cases the best strategy is to treat the medical
disorder or resolve substance use and not use psychiatric drugs to treat these
conditions. However, in some instances the use of antidepressants or
benzodiazepines may be indicated.
Adjunctive
Treatments
The anticonvulsant, Neurontin (gabapentin, daily doses:
300-2400 mg.) has significant antianxiety effects and has been found to be
useful in the treatment of generalized and social anxiety disorder. It has the
further benefit of being non-habit-forming and effective in relieving some
types of chronic pain (especially neuropathic pain). Additionally, antidepressants may exacerbate bipolar disorder. Rates of co-occurring bipolar and anxiety disorders are high. Therefor gabapentin may be an alternative. Finally, the drug Lyrica is approved to treat anxiety in Great Britain, and is sometimes used in the USA to treat anxiety, off- label. The over-the-counter
product, Kava Kava has antianxiety properties however it has been associated
with cases of liver toxicity and is not recommended for safety reasons.
Transcranial magnetic stimulation (with low frequency stimulation over the
right frontal lobe) has been used experimentally to treat PTSD and may be an
option for other types of anxiety disorders. Finally, exercise therapy has a
proven track record of effectiveness in treating most forms of anxiety
disorder.
Attention Deficit/Hyperactivity Disorder (ADHD)
Introduction
ADHD affects approximately 7% of children and 4% of adults. With neurological
maturation, most teenagers with ADHD will experience a noticeable reduction
in motoric restlessness/hyperactivity, but the core symptoms of ADHD (e.g. impulsivity,
impaired attention and lack of intrinsic motivation) continue through adolescence
and on into adulthood. Most experts agree that about one-third of ADHD children
completely outgrow the disorder by early adulthood (likely due to the ongoing
maturation of the prefrontal lobes which may continue until the late 20’s or
early 30’s). Two thirds experience ongoing symptoms throughout life.
Diagnostic Issues
It is very important
to emphasize that most psychiatric disorders in childhood present with some
degree of motoric restlessness and inattention. These outwardly observable behaviors absolutely do not
automatically lead to a diagnosis of ADHD.
The box below lists those disorders that must be considered in any
comprehensive evaluation of children or adolescents with hyperactivity and
inattention.
Differential
Diagnosis of Childhood Onset Psychiatric Disorders Presenting with Hyperactivity
and Inattention |
- Diffuse brain
damage (e.g. commonly seen in fetal alcohol syndrome or fetal exposure to other drugs of abuse)
- Post-Traumatic brain injuries
- Reactive attachment disorders
- Anxiety
disorders (e.g. separation anxiety)
- Post-traumatic stress disorder
- Agitated depression
- Situational
stress
- Bipolar mania
- Pre-psychotic
conditions
- Impaired affect
regulation associated with severe early abuse or neglect
- Boredom
(especially likely in bright children who are academically under-stimulated)
|
The
diagnosis of ADHD is based largely on three sources of data: family history (since ADHD is considered to
be a genetically transmitted disorder and thus often runs in families), a very
careful history detailing the nature and onset of behavioral symptoms, and a
description of current symptoms (especially as they vary across
situations). It is also a diagnosis of
exclusion (one must always first rule out those disorders listed above).
The
most common presentation for ADHD is an early onset (often present in infancy)
of restlessness, unstable sleep patterns, affective lability (especially,
crying a lot and difficulty in being soothed).
Most true ADHD children are identified in preschool when they have their
first sustained contact with other children and encounter social standards
(expectations to control behavior, follow rules, and stay on task in age
appropriate ways). There is some controversy among experts, but general
agreement that this very early onset of significant behavioral problems
is very characteristic of ADHD.
However, there is emerging data to suggest that some children destined
to have bipolar disorder may show early-onset behaviors that are ADHD-like
(i.e. prodromal symptoms of bipolar).
During
childhood the following symptoms predominate:
hyperactivity, impulsivity, impaired self-control, difficulties staying
on-task and limited ability for intrinsic motivation (e.g. motivation to stay
focused, especially on mundane, non-exciting or low stimulus-value tasks). Such symptoms are often highly
context-dependent; that is: most
noticeable in situations requiring that the child remain still and quiet (e.g.
in the classroom), yet may not be as noticeable when in an environment that is
inherently exciting, novel or stimulating (e.g. playing video games).
With
adolescence, as noted above, motoric hyperactivity often is reduced, but core
symptoms remain. Disorganization
(manifest by messy lockers, notebooks and bedrooms) often is pronounced in the
adolescent ADHD client (also true with adults suffering with ADHD), as are increasing problems adapting to society’s and
school’s demands for independent task performance and self-control.
It is important to note that the so-called “inattentive type” of ADHD (in DSM-5: ADHD with inattentive specifier) in many respects is a totally unrelated neurologic disorder (children with this disorder do show impaired attention; however, they are not hyperactive nor impulsive). This fundamental difference is also underscored by the failure of stimulants to effectively treat the inattentive subtype (it should be noted that most people without ADHD when given stimulants do show a degree of enhanced abilities to maintain attention…this can occur to a limited degree when used with children with the inattentive specifier, but the results are nowhere near as robust as seen in typical ADHD).). Currently there are no effective medication treatments for those suffering with the inattentive version of ADHD.
Neurobiology
Numerous
studies suggest impaired frontal lobe functioning in people suffering from ADHD
(evident in studies of metabolic functioning: e.g. SPECT and PET scans). In addition, abnormalities have been shown
in the dopamine neurotransmitter system.
Likewise, dopamine agonists (e.g. stimulants or bupropion) [Wellbutrin] are effective
medications for reducing ADHD symptoms. Minor structural abnormalities have
also been found in the brains of ADHD subjects (e.g. smaller cerebellar
volumes, smaller volumes of frontal and temporal areas and a smaller caudate
nucleus).
Appropriate
treatment with stimulants may not only reduce symptoms, but also may also
normalize the chemical microenvironment of the developing brain, and ensure
more normal brain maturation.
Castellanos, et al. (2002) demonstrated that ADHD children have smaller
cerebral and cerebellar volumes than age-matched controls. The degree of reduction in frontal,
temporal, cerebellar, and white matter volumes correlated significantly with
parent and teacher ratings of ADHD symptom severity. Unmedicated ADHD subjects exhibited strikingly smaller white matter
volumes compared to both controls and medicated ADHD children (treated with
stimulants). This suggests that
appropriate treatment may be neuroprotective.
Psychopharmacology
of ADHD
There are three classes of
medications with empirical support of efficacy in the treatment of ADHD: stimulants, certain antidepressants and a-2 adrenergic agonists.
Stimulants
The
mechanism of action of stimulants is inhibition of dopamine reuptake
(additionally, amphetamines promote increased release of dopamine from
vesicles). Listed below are the
currently available stimulants. There
are different ways to categorize stimulants, either by the onset of action or
duration of action. In general, most of
these agents have a moderately rapid onset, with symptom reduction occurring
30-45 minutes after ingestion, and a duration of action ranging from 4-12
hours. Depending on the formulation,
dosing is two to three times daily, with some long acting products providing
once daily dosing. What is most
important is to find the best possible dose and dosing schedule for a given
patient.
In the past few years there has been an explosion of various drugs that have received FDA approval, most of which are versions of methylphenidate. Listed below are those in most commonly in use.
Stimulants |
Immediate Release |
Typical Daily Doses |
- Methylphenidate
- Ritalin
- Metadate
- Methylin
- Dexmethylphenidate (Focalin)
|
10-60 mg
10-60 mg
10-60 mg
5-20 mg
|
|
5-40 mg
|
|
15-30 mg
|
- Amphetamines
- Amphetamine mixed salts (Adderall)
- Methamphetamine (Desoxyn)
- Amphetamine salts (Mydayis)
- Amphetamine sulfate (Adzenys)
Amphetamine salts (Evekeo)
|
5-40 mg
5-25 mg
12.5-50 mg
3.1-18.8 mg
5-40 mg
|
|
30-70 mg |
Extended Release |
Typical Daily Doses |
- Methylphenidate
- Ritalin SR
- Ritalin LA
- Metadate ER
- Metadate CD
- Methylin ER
- Concerta
- Quillivant XR (liquid)
|
20-60 mg
20-60 mg
10-60 mg
20-60 mg
20-60 mg
18-54 mg
10-60 mg
|
|
5-40 mg
|
|
5-40 mg
|
There
are over 200 well-controlled studies of stimulant use and outcomes are
significantly positive. Assuming an accurate diagnosis, any one stimulant taken
results in approximately 70% response rate (good to very good response). Although the stimulants are similar, there
are differences. Thus, if a trial with one stimulant (e.g. methylphenidate) is
less than optimal, then it is advisable to conduct a trial on another stimulant
(e.g. dextroamphetamine). If
systematic trials are conducted on each of the three classes of stimulants,
good outcomes are seen in about 90% of patients treated (Barkley, 2000). Across studies effect sizes are quite high, ranging from 0.8 to 1.0.
The
following briefly highlight important issues regarding stimulant treatment:
- Very low risk
of abuse in ADHD patients
- Side effects
- Initial insomnia (if taken later in the day)
- Solutions: earlier dosing, clonidine or trazodone given at bedtime
- Anorexia: generally, only affects the patient when the drug is active (i.e.
usually only interferes with appetite at lunch time). Has not been associated
with significant problems obtaining adequate nutrition. Focalin may have
less of this effect.
- Stomach ache
- Mild dysphoria
- Switch classes of stimulants
- Add an antidepressant such as bupropion
- Lethargy, sedation or impaired concentration: usually indicates that the
dose is too high
- ADHD affects all aspects of life; not just academic performance. Therefore, it
is often best to administer medications every day (i.e. not just on school
days)
- Start treatment with immediate release formulations and then switch to extended
release preparations if tolerated and effective
- Stimulants only work for a short period of time, and the positive effects
wear off in the late afternoon or evening. Thus, co-administration of antidepressants
(see below) may be an option for targeting symptoms later in the day
- Most children successfully treated with stimulants will require ongoing
medication treatment well into adolescence and possibly into adulthood
Failure to accurately diagnose and then to mistreat with stimulants can
have very adverse consequences (see table below).
Consequences of Misdiagnosis and Stimulant Treatment |
Diagnosis |
Consequences |
Anxiety disorder |
Increased anxiety |
Agitated depression |
Increased agitation |
Pre-schizophrenic |
Psychosis |
Bipolar disorder |
Increased manic symptoms
May cause cycle acceleration |
Situational stress |
Failure to address psychological issues |
Alpha-2 Adrenergic Agonists
Clonidine (Catapres; Kapvay) and guanfacine (Tenex; Intuniv) may be used to treat core ADHD
symptoms (see figure below), however they are most effective in reducing irritability,
aggression, and impulsivity, and promoting sedation (to treat initial insomnia).
Alpha-2 agonists are also the treatment of choice for comorbid tics.
Alpha-2 Adrenergic Agonists |
Generic Name |
Brand Names |
Typical Doses |
clonidine |
Catapres; Kapray |
0.15-0.4 mg (1) |
guanfacine |
Tenex; Intuniv |
0.25-1.0 mg (2) |
(1) Three to four times a day
(2) Two to three times a day |
There have been 25 reported cases of death in children taking clonidine in
conjunction with a stimulant (USA and Canadian data combined). However, the FDA has conducted an investigation
and failed to find any significant cause for concern in co-administering these
drugs. The deaths seen, as determined upon autopsy, occurred in children who had evidence of pre-existing, yet un-diagnosed significant cardiac disease. It is prudent to do cardiac screening on all subjects prior to administering the medications, including a family history of early heart disease, sudden infant death syndrome, a history of fetal alcohol exposure, or any of the following symptoms: sudden, unexplained loss of consciousness; dizziness; tachycardia; or, chest pain. Should any of these exist it is recommended that the child be given a pre-treatment EKG. Combined use of Alpha-2 agonists and stimulants is a common practice
(both for treating ADHD and comorbid ADHD and tics) (Walkup, 2003).
Antidepressants
20% of ADHD children
will experience co-occurring depression.
Antidepressants certainly may be helpful in reducing mood symptoms. However, beyond this use of antidepressants,
certain classes of antidepressants have been shown to have positive effects on
core ADHD symptoms. Not all antidepressants treat ADHD; only those that
increase the availability of dopamine or norepinephrine (thus SSRIs, although
often a good adjunct for treating anxiety or depression, are not effective in
treating core ADHD symptoms). Antidepressants
that have evidence of efficacy in treating ADHD are listed below.
Antidepressants Used to Treat ADHD |
Generic Name |
Brand Name |
Typical Daily Dosage |
bupropion |
Wellbutrin SR/LA |
150-300 mg |
atomoxetine |
Strattera |
1.2-1.8 mg/kg |
Treatment outcomes with
antidepressants are not as robust as that seen with stimulants, however they
afford several advantages:
- Once a day
dosing
- No need for
triplicate prescription
- No addiction
potential
- Effects
(generally seen within 5-40 days after initiating treatment) typically last
24-hours a day
- Can treat comorbid depression
Appendix
Psychiatric
Medications: Quick Reference Guide
To keep up-to-date with new medications, click on my website www.Psyd-Fx.com for a free download of the Psychiatric Medications Quick Reference Chart.
This appendix may be copied and given to patients who desire detailed information
regarding their medications.
Note: To the best of
our knowledge doses and side effects listed in the Quick Reference Guide, linked to below, are accurate. However, this
is meant as a general reference only and should not serve as a guideline for
prescribing medications. Please check the manufacturer’s product information sheet or the P.D.R. for any changes in dosage schedule or contraindications. (Brand names are registered trademarks.)
Mood Stabilizers/Anti-manic agents
Lithium
Lithium Facts:
- the drug with
the best track record in preventing recurrences of episodes.
- lithium is a
naturally occurring element
- lithium is the
only psychiatric drug proven to substantially reduce the incidence of suicide
Brand Names:
Lithium carbonate: Lithotabs, Eskalith,
Lithonate, Lithane, Carbolith, Lithobid, Duralith
Lithium citrate:
Cibalith
Uses: Treats mania,
bipolar depression and is used to reduce recurrences of mania and depression.
Used to augment antidepressants in treating major depression
Typical Adult Daily
Doses (Eskalith or Lithonate: most commonly prescribed): 600-2400 mg. per day.
Note: what matters with lithium treatment is not the dose, per se, but the
blood level (which is carefully monitored). A lithium level between 0.8 and 1.2
mEq/l (mEq/l is the technical designation for what is commonly called the
lithium level) is generally felt to be in the therapeutic range for treating
mania. Once the manic episode is resolved, then it is common practice to lower
the dose to establish a blood level somewhere between 0.6 and 0.8 mEq/l. Blood
levels above 1.2 are associated with significant side effects, and levels above
2.0 can be dangerous.
Onset of effects
(how long it takes to start working): generally, 7-10 days
Laboratory Tests:
Prior to starting
treatment with some medications, laboratory tests are required to establish
baseline measures of the functioning of certain bodily systems. The following
are typically required prior to starting treatment (and those with an * will
need to be monitored periodically during treatment).
ECG (EKG)*,
electrolytes, complete blood count, kidney function tests* (BUN, creatinine,
urinalysis), thyroid tests*, calcium*, pregnancy test (optional).
Laboratory Tests routinely done in an ongoing basis:
Those tests above
flagged with an asterisk are repeated periodically. In addition, it is necessary
to periodically check lithium blood levels. This is done frequently during the
first weeks of treatment and when there are significant changes in dosage. Once
a person is stabilized on lithium for several months, lithium levels will then
be checked less often (e.g. 3-4 times a year).
Common Side Effects:
- Nausea or heartburn
- Muscle tremor or
weakness
- Decreased sex drive
- Lethargy, drowsiness
(may impair the ability to safely drive an automobile)
- Difficulty
concentrating
- Weight gain
- Increased thirst and
frequency of urination
- Rash or acne
Less Common Side
Effects
- Loss of balance
- Double vision
- Vomiting
- Diarrhea
- Slurred speech
- Trembling
Rare or potentially
dangerous (require emergency medical treatment)
- Soreness of mouth,
throat or gums
- Severe rash or
itchiness
- Swelling of the neck
or face
- Severe: nausea,
vomiting, weakness, fever, flu-like symptoms
- Marked increase in
thirst and very frequent urination
Habit-Forming /
Addiction Potential: none
Interactions with
other medications:
Here we will list only the most
common medications with which the drug may cause adverse
interactions.
- diuretics (water
pills)
- calcium channel
blockers
- ACE inhibitors
- Non-steroidal
anti-inflammatory pain medications such as ibupropin
- theophyline
Safety during
pregnancy: lithium is generally considered to be safe for use during pregnancy,
however there is a slight risk for a rare birth defect
(Ebstein’s anomaly, a heart defect) if taken during the first
trimester. This occurs 0.1-0.2% of fetuses exposed to
lithium.
Breast feeding: not
recommended when taking lithium
Special Concerns
Lithium is a very dangerous drug if taken in an accidental or intentional overdose.
In the event of an overdose the patient must seek immediate medical attention. Also, lithium blood
levels can become dangerously high if one experiences marked dehydration (e.g. commonly
seen if there is an illness that causes severe vomiting and diarrhea).
Anticonvulsant Mood Stabilizers
Anticonvulsant
Facts
Anticonvulsants are medications originally developed to treat epilepsy. It was only by accident that it was discovered that some anticonvulsants also have the ability to treat mania. In addition the anticonvulsant Lamictal has antidepressant actions and can be used to treat bipolar depressive episodes.
Anticonvulsant Mood
Stabilizers:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
divalproex |
Depakote |
750-1500 mg |
| carbamazepine |
Tegretol; Equatro |
600-1600 mg |
| oxcarbazepine |
Trileptal |
1200-2400 mg |
| lamotrigine |
Lamictal |
50-200 mg |
An additional anticonvulsant, tiagabine (brand name Gabitril) is considered to be an experimental treatment. There are positive case reports of the use of Gabitril for the treatment of mania, but to date no well-controlled research studies.
Uses: Treat mania. Lamictal is used to treat bipolar depression. Research evaluating
the ability for anticonvulsants to help prevent recurrences of mania and bipolar
depression is not yet conclusive. Depakote, Trileptal and Tegretol likely help
to prevent recurrences of mania and Lamictal likely reduces the recurrence of
bipolar depression.
Therapeutic blood
levels: two of the anticonvulsant mood stabilizers must be periodically monitored to
check the levels of medication present in blood.
| Depakote blood levels: |
50-125 mcg/ml |
| Tegretol or Equatro blood levels: |
4-12 mcg/ml |
| Trileptal blood levels: |
not yet established |
| Lamictal blood levels: |
not necessary to monitor |
Onset of effects:
generally, 7-10 days (one exception: if high doses of Depakote are administered,
effects can be seen in four days)
Laboratory Tests:
Required for: Depakote, Tegretol, Equatro and Trileptal. Specific tests depend
on which drug is used, but often include the following:
Complete blood
count, platelets, electrolytes, cholesterol, triglycerides, sonogram of ovaries
(optional: for females under the age of 20 treated with Depakote), liver
function tests, ECG (EKG), pregnancy test. For Topamax: kidney function tests
(BUN and creatinine).
Pretreatment labs are generally not required for Lamictal
Laboratory Tests
routinely done on an ongoing basis:
Tegretol, Equatro, Depakote and Trileptal blood levels must be monitored (especially
during the initial weeks of treatment). Generally, once a person is stabilized
on Depakote or Trileptal, blood level monitoring is not necessary. However,
those treated with Tegretol or Equatro must have periodic and ongoing monitoring
of blood levels.
Ongoing lab test are generally not required for Topamax or Lamictal
Common Side Effects
(each medication has specific side effects, however listed here are side
effects that can be seen in most of the anticonvulsants).
- Drowsiness, lethargy
- Mild dizziness
- Unsteadiness when
standing or walking
- Difficulty
concentrating
- Blurred vision
- Dry mouth
- Muscle tremor
- Nausea or heartburn
- Weight gain (can
occur in all except Topamax)
- Weight loss: Topamax
- Changes in menstrual
cycle
- Decreased sex drive
Less Common Side
Effects
- Infertility problems
(seen with some women under the age of 20 treated with Depakote), also
accompanied by menstrual irregularities.
- Changes in hair:
hair loss (usually transient) or changes in hair texture
- Rash or itching
Rare or potentially
dangerous (if these occur patient should seek emergency medical care)
- Skin rash (mild rashes are fairly common, but a rash that is severe and
rapidly increases in severity should be reported to your doctor immediately)
- Confusion
- Yellow tint to skin
or eyes
- Severe problems with
balance and dizziness
- Soreness of mouth,
throat or gums
- Easily bleeding or
bruising
- Severe nausea,
vomiting, and abdominal pain
Habit-Forming /
Addiction Potential: none
Interactions with
other medicines (varies depending on the specific drug):
- Birth control pills (especially with Tegretol or Equatro; may occur with
Topamax)
- Anticoagulants
(blood thinners)
- Aspirin (moderate to
high doses)
- Cimetidine (Tagamet)
- Antibiotics (especially with Tegretol or Equatro)
- Calcium channel
blockers
- Propoxyphene (Darvon) (especially with Tegretol and Equatro)
- Antidepressants
(anticonvulsant mood stabilizers are often prescribed for patients who are also
taking antidepressants…generally there are no problems unless the
antidepressant doses are high)
- Antacids: may affect
the absorption of some anticonvulsants
Safety during
pregnancy: there is a risk of birth defects when taking anticonvulsant mood stabilizers
during pregnancy (especially the first trimester). Among the anticonvulsants Depakote is the most dangerous drug to use during pregnancy. Most psychiatrists do not
prescribe these medications during pregnancy.
Breast feeding: not
recommended when taking anticonvulsants
Antipsychotic Medications
Antipsychotic
Medication Facts
Antipsychotic
medications were first developed to treat psychotic symptoms such as
hallucinations. The first such drugs were found to be effective in reducing
psychotic symptoms, but they were notoriously “dirty” drugs, causing
significant side effects. Since the mid-1990s new and improved antipsychotics
have been developed and marketed. These newer generation medications are not
side effect-free, but they are considerably safer and better tolerated. The
newer drugs are commonly referred to as atypical antipsychotics.
Although atypical
antipsychotic medications are highly effective in treating psychotic symptoms
it has been found that they are also good treatments for mania, to augment
treatments for depression, to treat aggression and possibly for mood stabilization
in general.
Atypical
(SGA: second generation) Antipsychotic Medications:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
olanzapine |
Zyprexa |
5-20 mg |
| risperidone |
Risperdal |
4-10 mg |
| paliperidone |
Invega |
4-12 mg |
| ziprasidone |
Geodon |
60-160 mg |
| aripiprazole |
Abilify |
15-30 mg |
| quetiapine |
Seroquel |
150-400 mg |
| asenapine |
Saphris |
10-20 mg |
| iloperidone |
Fanapt |
12-24 mg |
| lurasidone |
Latuda |
40-80 mg |
| brexpiprazole |
Rexulti |
1-4 mg |
| cariprazine |
Vraylar |
1.5-6 mg |
Another
antipsychotic medication that is used occasionally is clozapine (generic),
Clozaril (brand name). This
older-generation antipsychotic medication has significant side effects (e.g.
dry mouth, constipation, sedation, seizures, excessive salivation, blurred
vision, nausea, heartburn and weight gain) and has been associated with a
serious blood disorder (agranulocytosis: which causes soreness of the mouth,
throat and gums and a high fever). For this reason, it is never considered to be a first
line medication choice. However, despite the problematic side effects, Clozaril
is an important medication that can often successfully treat those rare people
who have not responded to more traditional mood stabilizers, antipsychotics or
antidepressants. The typical adult daily doses for Clozaril are: 300-900 mg.
Please note that all of the following information regarding antipsychotics pertains to the atypical antipsychotics or SGAs (with the exception of Clozaril).
Uses: Treat mania
and agitation; treat psychotic symptoms associated with schizophrenia
and both manic and depressive episodes; treat treatment-resistant
schizophrenia, bipolar and major depression. The effectiveness of
antipsychotic medications in the long-term prevention of
recurrences of bipolar illness is as yet inconclusive.
Onset of effects:
Antipsychotic medications used to treat mania or acute psychosis and can begin
to reduce severe agitation within a few hours to a few days, however, the
reduction of more pronounced manic and psychotic symptoms, is similar to that
seen with other mood stabilizers such as lithium and anticonvulsants and other
antipsychotics (7-10 days or longer).
Laboratory Tests: It is important to monitor triglycerides and cholesterol levels and blood glucose levels because many of these drugs can cause or aggravate type II diabetes. Weight gain must also be monitored. These lab tests are indicated in all new generation / atypical antipsychotics except Abilify and Geodon (it appears that the newest antipsychotics, Saphris, Fanapt and Latuda, may have a low incidence of metabolic side effects). Also frequent blood tests are required for those taking Clozaril to monitor for possible agranulocytosis. The blood monitoring must be done once a week for the first 6 months, during the next 6 months, every other week, and thereafter, once a month.
Common Side Effects
for Atypical Antipsychotics (SGAs):
- Drowsiness and
lethargy (can occur with all, except Abilify)
- Weight Gain (can
occur with all, however minimal with Geodon and unlikely with Abilify)
- Nausea, vomiting,
heartburn
- Stuffy nose
- Mild dizziness
Less Common Side
Effects
- Constipation
- Decreased Sex Drive
- Breast tenderness,
liquid discharge from breasts (can occur with high doses of Risperdal)
- Rash
- Severe sunburn when
exposed to even moderate amounts of sunlight
- Muscle rigidity
- Tremor
- Rash
- Blurred vision
- Abnormal,
involuntary movements of the mouth, tongue and sometimes head, neck and hands
(rare)
Rare or Potentially
Dangerous Side Effects (if these occur, the patient should seek emergency medical care)
- Soreness
in mouth, throat and gums
- Moderate to severe
nausea, vomiting and flu-like symptoms
- Yellow tint to skin
or eyes
- Seizures
- High fever
(especially if accompanied by muscle stiffness)
- Confusion
- Type II diabetes
- Increased
cholesterol or triglycerides
- Extreme restlessness
- Unusual bruising or
bleeding
- Severe rash
- Frequent urination
or loss of bladder control
Habit-Forming / Addiction Potential: none
Interactions with
other medications (varies depending on the specific drug)
- levodopa: decreases
effectiveness of levodopa
- amphetamines
- anticonvulsants: can
decrease antipsychotic blood levels
- Digoxin: may
increase blood levels of digoxin and the antipsychotic
- Warfarin: may
increase Warfarin blood levels
- Antacids: may
interfere with the absorption of the drug
Safety during
pregnancy: atypical antipsychotics are generally considered to be safe
during pregnancy
Breast-feeding:
antipsychotic medications are secreted in breast milk. Since these are recently developed
medications, there is inadequate information regarding safety to
infants.
Special Concerns:
- Avoid exposure to
extreme heat (e.g. saunas)
- Grapefruit juice may
interfere with the effects of the drug
- Excessive cigarette
smoking may interfere with the effects of the drug
- Avoid direct
exposure to sunlight: antipsychotic medications can increase the likelihood of
severe sunburns
Antidepressant Medications
New Generation
Antidepressants:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
| trazodone |
Desyrel |
50-400 mg |
fluoxetine |
Prozac, Sarafem |
20-80 mg |
bupropion |
Wellbutrin |
150-400 mg |
sertraline |
Zoloft |
50-200 mg |
paroxetine |
Paxil |
20-50 mg |
venlafaxine |
Effexor |
75-350 mg |
nefazodone |
Generic only |
100-500 mg |
| fluvoxamine |
Luvox |
50-300 mg |
mirtazapine |
Remeron |
15-45 mg |
citalopram |
Celexa |
10-60 mg |
escitalopram |
Lexapro |
5-20 mg |
duloxetine |
Cymbalta |
20-80 mg |
atomoxetine |
Strattera |
60-120 mg |
| desvenlafaxine |
Pristiq |
50-400 mg |
| vilazodone |
Viibryd |
10-40 mg |
| vortioxetine |
Trintellix |
10-20 mg |
| leyomilnacipran |
Fetzima |
40-120 mg |
Antidepressants (all
except Wellbutrin) are effective in treating severe anxiety, panic attacks and
obsessive-compulsive disorder (OCD).
Uses: Treat unipolar major
depression, dysthymia, bipolar depression, anxiety disorders, and
obsessive-compulsive disorder (OCD), bulimia
Onset of Effects:
generally, 2-6 weeks
Laboratory Tests:
not required
Common Side Effects:
- Nausea, heartburn
- Energized or anxious
feelings (typically subsides within 1-2 weeks)
- Headaches
- Sedation (primarily
with Remeron and Desyrel)
- Difficulty falling
asleep (often subsides in a few weeks)
- Sexual dysfunction:
primarily inorgasmia (difficulty achieving an orgasm despite adequate arousal)
(can occur in 14-30% of people treated with antidepressants. One exception:
very rare with Wellbutrin)
- Weight gain
(primarily with Remeron; with other antidepressants, weight gain can occur in
up to 10% of people, however the weight gain typically does not occur until the
person has been taking the drug for a period of time longer than six months)
- Muscle tremor
- Rash
Rare Side Effects
(if these occur, the patient should seek emergency medical care)
- Soreness of mouth,
throat, or gums
- Severe rash
- Seizures
- Unusual bruising or
bleeding
- Severe nausea,
vomiting and flu-like symptoms
- Severe agitation or
restlessness
- Yellow tinge to skin
or eyes; dark colored urine
- Rapid shift into
mania or hypomania; racing thoughts
Habit-Forming / Addiction Potential: none
Interactions with
other medications: (varies depending on the drug)
- Do not take with St. John’s Wort,
5-HTP (dietary supplement), MAOIs, cimetidine (Tagamet)
Safety during pregnancy: most experts agree that some new generation antidepressants
are relatively safe for use during pregnancy (e.g. Prozac, Celexa, Lexapro, Zoloft, Effexor,
Wellbutrin and Luvox). Paxil is contraindicated during pregnancy. (Note: the following antidepressants have only recently
come to market and there is inadequate data to evaluate safety during pregnancy:
Cymbalta, Strattera, Lexapro, Celexa, Nefazodone and Remeron). High doses
of Desyrel (trazodone) should not be used during pregnancy.
Breast-feeding:
antidepressants are secreted in breast milk, but the amounts are extremely low.
Most experts agree that it is safe to breast feed while taking new generation
antidepressants.
Special Concerns: If
a patient has been taking antidepressants for a period of six weeks or more,
abruptly stopping the medications, there can be withdrawal symptoms (this
can occur with any of the antidepressants with the exception of Prozac).
Withdrawal symptoms include: nausea, stomach upset, nervousness, and flu-like
symptoms. Withdrawal symptoms are very unlikely if you have been taking the
medication for less than six weeks. And withdrawal symptoms can be avoided
almost 100% of the time by reducing the dose gradually.
Antianxiety Medications: Benzodiazepines
Benzodiazepine
Facts:
Benzodiazepines are
also commonly referred to as minor tranquilizers or anti-anxiety medications.
Benzodiazepines:
Generic Name |
Brand Name |
Typical Adult Daily Dose |
| diazepam |
Valium |
4-30 mg |
| clonazepam |
Klonopin |
0.5-2.0 mg |
| lorazepam |
Ativan |
2-6 mg |
| oxazepam |
Serax |
30-60 mg |
| alprazolam |
Xanax |
1-4 mg |
Benzodiazepine and benzodiazepine-like medications used
for sleep:
Generic Name |
Brand Name |
Typical Adult Nighttime Dose |
| temazepam |
Restoril |
15-30 mg |
| triazolam |
Halcion |
0.25-0.5 mg |
| estazolam |
Prosom |
1-2 mg |
| zolpidem |
Ambien |
5-10 mg |
| zaleplon |
Sonata |
5-10 mg |
| eszopiclone |
Lunesta |
1-3 mg |
Uses: Treats acute
anxiety, agitation and insomnia in acute mania, anxiety disorders (such as
panic disorder, post-traumatic stress disorder and generalized anxiety
disorder).
Onset of Effects:
30-45 minutes
Laboratory Tests:
none required
Common Side Effects:
- Drowsiness
- Dizziness
- Forgetfulness
- Slurred speech
Less Common Side
Effects:
- Confusion
- Nervousness
- Rash
- Loss of balance and falls
Habit-Forming / Addiction Potential: Significant risk for people with a prior
personal or family history of alcoholism or other forms of serious drug abuse.
Currently, Lunesta has been shown to be non-habit-forming. It is relatively
new and as with all new drugs, the final evaluation will unfold as the medication
has been on the market for a while.
Interactions with
Other Medications: When taking benzodiazepines or any other type of medication
that causes drowsiness or impaired alertness and reaction time can be
potentially dangerous, especially if one has to drive and automobile. In
addition, alcohol should not be consumed when taking benzodiazepines.
Safety During
Pregnancy: Benzodiazepines typically are not to be used during pregnancy.
Breast Feeding:
Benzodiazepines are secreted in breast milk and should not be used when
breast-feeding.
Special Concerns: If benzodiazepines are being taken on a
regular basis the body develops a tolerance for the medication. When this
happens, typically the drugs continue to work to reduce anxiety, but the
problem is that when there is tolerance, if abruptly stopped, there can be withdrawal symptoms. Withdrawal symptoms usually include nervousness,
agitation, difficulty falling to sleep, and on occasion can produce seizures.
This needs to be taken very seriously. If a patient has been taking a benzodiazepine
on a daily basis for more than 6 weeks and especially if the dose is moderate
to high, withdrawal reactions are a very real risk. Never abruptly
stop taking the medication except under medical supervision. It is
also a good idea for patients to be especially careful to monitor their supply of the
medications so that refills can be requested in a timely fashion. Many people
find it helpful to keep at least a two-day supply on hand in the event that it
takes longer than usual for a prescription to be refilled.
Atypical Sleeping Medications
Generic Name |
Brand Name |
Typical Adult
Nighttime Dose |
Description |
| ramelteon |
Rozerem |
4-16 mg |
Targets melatonin receptors |
| doxepin |
Selenor |
3-6 mg |
A sedating low dose antidepressant marketed as a sleeping pill |
| suvorexant |
Belsomra |
15-40 mg |
Targets orexin receptors: a different mechanism than Benzodiazepines |
Note: Over-the-counter Melatonin in doses of 2-10 mg have been used for years to treat initial
insomnia. Recently it has been found that these higher doses may exacerbate depression
and destabilize bipolar disorder. Low dose melatonin (0.5 mg) may enhance the quality
of sleep, but it does not produce sedation. It remains to be seen if a low dose strategy is
helpful and safe for use with mood disorders.
Calcium Channel Blockers
Calcium channel blockers are medications that are often used to treat certain
cardiovascular diseases. One of these drugs has been found to be effective in
the treatment of mania, and possibly as a treatment to prevent the recurrence
of mood episodes.
Generic Name |
Brand Name |
Typical Adult Daily Dose |
| verapamil |
Calan, Isoptin |
120 mg. given 3 or 4 times a day
Thus total daily: 360-480 mg |
Uses and General
Considerations: for people who cannot tolerate lithium, for rapid cycling
bipolar, or for use during pregnancy (verapamil is considered to be the safest
mood stabilizing medication for the treatment of bipolar disorder during
pregnancy). Like most other mood stabilizers, it generally takes 7-10 days to
begin reducing symptoms.
One additional
calcium channel blocker is used occasionally to treat mania: nimodipine (brand
name: Nimotop). This medication looks promising in terms of efficacy; however, it
is very expensive and to date although there are positive case reports, there
are no well controlled studies.
Miscellaneous
Omega-3 Fatty Acids
During the past ten years a number of studies have been conducted with people suffering from bipolar disorder and also major depression. Preliminary findings strongly suggest that adding omega-3 fatty acids to the diet can have a positive effect on reducing unipolar depressive symptoms when added to antidepressant medications in some individuals. Results in bipolar disorder have been minimal and disappointing. The best source of omega 3 fatty acids is fish oil because it has better bioavailability in the brain versus omega 3 from nut and seed oil.
Uses and General Considerations: Omega-3 fatty acids: Studies have found that people treated with omega-3 fatty acids must take these dietary supplements on a daily basis and over a prolonged period of time (i.e. building this into one's ongoing diet). Doses recommended for treating depression are 1000-2000 mg per day. The version of omega-3 that has effects on mood is EPA.
Older Generation Antipsychotics
As noted above,
newer generation antipsychotics have been developed during the past ten years.
The newer drugs are considerably safer and have significantly fewer side
effects than older-generation antipsychotic medications. We are simply
mentioning these medications here just as a point of information since in rare
instances some people may be treated with these drugs (brand names): Thorazine,
Mellaril, Serentil, Trilafon, Loxitane, Stelazine, Prolixin, Navane,
Orap, and Haldol. Of these the most common drugs that are still used these days
are Haldol (often useful to initially treat very severe agitation seen in some
types of mania or schizophrenia) and Trilafon.
Anticholinergic Medications
This class of
medications is used occasionally to combat side effects of some antipsychotic
drugs (side effects such as: muscle rigidity or spasms, restlessness, tremor).
Again, we will only list these medications (brand names): Cogentin, Akineton,
Artane. Anticholinergic drugs have their own set of side effects including:
constipation, blurred vision, dry mouth, difficulty beginning urination, and
occasionally memory loss, confusion and delirium.
SAM-e
SAM-e
(S-adenosylmethionine) is a naturally occurring bio-molecule found in most
living cells. It is felt to be necessary for carrying out a number of important
intracellular chemical reactions. SAM-e has been used in Europe for more than
20 years as a treatment for depression. A number of studies have shown it to be
equally effective when compared to prescription antidepressants. Most notable
is the virtual lack of side effects. It has not been well supported in studies
of the treatment of bipolar disorder, however it may prove to be a viable
option for treating bipolar depression. It, however, is not useful for treating
mania and has, in fact, been found to switch people with bipolar depression
into states of mania. Doses for the treatment of depression range from 400-1600
mg per day, although recent investigations indicate that often-higher doses
(1200-1600 mg per day) may be necessary for effectively reducing depressive
symptoms. This is available over-the-counter (i.e. not requiring a
prescription). It is recommended that if you are considering using SAM-e,
it should only be taken with close observation by your treating psychiatrist.
St. John’s Wort
St. John’s Wort is
an over-the-counter dietary supplement that has been found to have
antidepressant properties. A meta-analysis (Linde, et al., 2008) has shown that
St. John’s Wort is equally effective to prescription antidepressants in the
treatment of depression if taken in large doses (e.g. 1800 mg per day). This herbal remedy is generally well
tolerated with few if any side effects. There are reported cases of possible
infertility problems associated with its use, although it is as yet unclear
whether this is a common side effect. St. John’s Wort requires daily dosing of
900 (mild depression) -1800 mg. per day (taken in three divided doses, for major depression), and typically the first
signs of symptom improvement take about six weeks to emerge. Thus, the onset of
action is longer than that seen with prescription antidepressants. Research on
the treatment of bipolar depression is sorely lacking. And as with any other
treatment that has antidepressant properties, St. John’s Wort can potentially
provoke mania. Caution: although St. John’s Wort, when it is the only medication
being taken, appears to be quite safe, it has been found to cause very
significant drug-drug interactions. It is strongly advised that patients never
take St. John’s Wort without first consulting with their doctor. Given its
tendency to cause multiple and complex interactions with a host of medications,
it is recommended that it not be used by people being treated with bipolar
medications.
Caffeine Questionnaire
Sleep Disturbances: A high yield treatment intervention often overlooked in the treatment of psychological disorders. www.PsyD-fx.com.
Following is a caffeine questionnaire that allows clinicians to quickly determine caffeine intake in their clients. Even small amounts of caffeine exacerbate anxiety symptoms and the ideal caffeine levels for those with situational stress and anxiety disorders is zero. Small amounts of caffeine taken in the morning may actually be helpful for people suffering from depression (if not otherwise contraindicated due to medical conditions).
Everyone knows that caffeine ingested close to bed time may interfere with the ability to go to sleep (i.e. initial insomnia). However, the more problematic effect of caffeine is its tendency to disrupt deep sleep (also known as slow wave or restorative sleep). High amounts taken in the morning (e.g. 750 mg+) and modest amounts of caffeine taken after noon may interfere with the ability to get adequate deep sleep, even if not causing initial insomnia. Caffeine use among people suffering from almost any type of psychiatric disorder often exceeds levels that are acceptable. Preserving deep sleep is essential for emotional and cognitive functioning. Our patients may not realize or appreciate the negative impact of excessive caffeine use. Sleep is essential for normal brain functioning and needs to be optimized in each and every client we see for treatment.
It is important to tell all clients about the need for good quality sleep and keeping caffeine at acceptable levels is a high yield intervention. Yet, many of our clients may minimize the importance of this, so good patient education is important. Generally speaking, for those with significant psychological disorders, caffeine levels should not exceed 250 mg per day and should not occur after noon (in order to preserve deep sleep). Afternoon fatigue is often easily addressed by a 10-minute brisk walk, which generally results in 60-120 minutes of energy and relief from fatigue. Daily use of caffeine exceeding 750 mg will require gradual caffeine reduction to avoid caffeine withdrawal (e.g. reducing daily use by 125 mg per day per week).
Please feel free to make copies of the following caffeine questionnaire for use with your clients.
Click here for the printable Caffeine Questionnaire
Suggested Readings/Resources
Advoka, C, (2018) Julien’s Primer of Drug Actions. Worth Publishers
bipolarnews.org. a free quarterly publication
Carlat Psychiatry Reports: a monthly subscription. Reports for adult psychiatry and one for children and adolescents. PO Box 626, Newburyport, MA
Geller, B. and DelBello, M.P. (Eds.) (2003) Bipolar Disorder in Childhood and Early Adolescence. The Guilford Press: New York.
Preston, J. and Johnson, J (2019; 2020 update: release date 1-1-2020)
MedMaster, Miami, Florida
Preston, J.D., O'Neal, J. and Talaga, M. (2017) Handbook of Clinical Psychopharmacology for Therapists: Eighth Edition. New Harbinger: Oakland.
Preston, J.D., O’Neal, J., and Talaga, M. (2015) Childhood and Adolescent Psychopharmacology Made Simple. Third Edition. New Harbinger: Oakland
Preston, J.D., O’Neal, J, and Talaga, M. (2009) Consumer’s Guide to Psychiatric Drugs. Second edition: Penguin Books: New York
Stahl, S. (2017) Prescriber’s Guide 6th edition, Cambridge University Press: Cambridge
References: Psychopharmacology
Akiskal, H.S. and Cassano, G.B., editors (1997) Dysthymia and the Spectrum of Chronic Depression. New York: Guilford Press.
Akiskal, HS, Bourgeois, M.L., et al. (2000) Re-evaluating the prevalence of and diagnostic composition within the broad clinical spectrum of bipolar disorders. J. Affective Disorders, 59, Suppl: S5-530.
Alarcon, R.D., et al. (2000) proposing an algorithm for the pharmacological management of PTSD. Annals of Clinical Psychiatry. 12(4): 239-246.
Barkley, R.A. (2000) Taking Charge of ADHD. New York: Guilford Press.
Bauer, M., et al. (2003) Implications of the neuroprotective effects of lithium
for the treatment of bipolar and neuro-degenerative disorders. Pharmacopsychiatry.
36 (3): S250-254.
Benes, F.M., et al. (2001) The density of pyramidal and non-pyramidal neurons in anterior cingulate cortex of schizophrenic and bipolar subjects. Biological Psychiatry. 50 (6): 395-406.
Biederman, J. et al. (1999) Pharmacotherapy for ADD reduces risk for substance
abuse disorder. Pediatrics. 104(2): e20.
Blehar, M.C. and Rosenthal, N.E. (1989) Seasonal affective disorders and phototherapy. Archives of General Psychiatry. 46 (5): 469-474.
Bowden, C.L., Calabrese, J.R., Sachs, G.S., et al. (2004) A placebo-controlled 18-month study of lamotrigine and lithium maintenance treatment in recently depressed patients with bipolar I disorder. Arch Gen Psychiatry. In press.
Calabrese, J.R., Bowden, C.L., Sachs, G.S., et al. (1999) A double-blind, placebo-controlled study of lamotrigine monotherapy in outpatients with bipolar I depression. J Clin Psychiatry. 60: 78-88.
Castellanos, F.X., Lee, P.P., et al. (2002) Developmental trajectories of brain volume abnormalities in children and adolescents with ADHD. JAMA. 288(14): 1740-1748.
Cooper, J.R., Bloom, F.E., and Roth, R.H. (2003) The Biochemical Basis of Neuropharmacology. Oxford: Oxford University Press.
Davis, L.L., et al (2001) Pharmacotherapy for PTSD: A comprehensive review.
Expert Opinion: Pharmacotherapy. 2(10): 1583-1595.
Dawson, R. et al. (1999) Course of treatment received by depressed patients. Journal of Psychiatric Research. 33: 233-244.
Delgado, P.L., Charney, D.S., Price, L.H., Aghajanian, G.K. Landis, H., and Heninger, G.R. (1990) Serotonin function and the mechanism of antidepressant action: Reversal of antidepressant induced remission by rapid depletion of plasma tryptophan. Archives of General Psychiatry, 47: 411-418.
Delle, C.R., et al. (2002) Efficacy and tolerability of oral and IM SAM-e in the treatment of major depression: comparison with imipramine in 2 multi-center studies. Am. J. of Clinical Nutrition. 76: 1172S-1176S.
Doraiswamy, P. M., Kahn, Z.M., Donahue, R.M., and Richard, N.E., (2001) Quality
of life in geriatric depression: A comparison of remitters, partial responders,
and nonresponders. American Journal of Geriatric Psychiatry, 9: 4223-428.
Doucherty, J.P. (1997) Barriers to the diagnosis of depression in primary care. J Clin Psychiatry. 58 (suppl 1): 5-10
Dubovsky, S.L. (2001) New concepts in the treatment of mania. U.C. Davis Symposium, Sacramento.
Echols, J., et al. (2000) SAM-e. Harvard Review of Psychiatry: 8, 84-90.
Fava, M. (2000) New approaches to the treatment of refractory depression. J Clin Psychiatry. 61 (suppl 1): 26-32.
Frank, E., Hastada, S., et al. (1997) Insuring lifestyle regularity in recovering bipolar disorder patients. Biological Psychiatry. 41: 1165-1173.
Geller, B. and DelBello, M.P. (Eds.) (2003) Bipolar Disorder in Childhood and Early Adolescence. The Guilford Press: New York
George, M.S. (2003) Stimulating the brain. Scientific American. 289 (3): 66-73.
Ghoemi, S.N., et al. (2001) Effectiveness and safety of long-term antidepressant treatment in bipolar disorder. J Clin Psychiatry. 62(7): 565-569.
Gitlin, M. (2002) Depression Myths and Contrary Realities. Continuing Education Series, UCLA.
Gitlin, M. (2002) Recognition and Resolve: Treatment Resistant Depression. U.S. Psychiatric Congress, Las Vegas.
Goodwin, F.K., Jamison, K.R. (2007) Manic-Depressive Illness. Second Edition New York:
Oxford University Press Inc.
Heim, C. and Nemeroff, C.P. (2002) Neurobiology of early life stress. Seminars in Clinical Neuropsychiatry. 7(2): 147-159.
Hirschfeld, R.M.A., et al. (2002) Practice Guidelines for patients with Bipolar Disorder. Washington, D.C. American J. Psychiatry, 159, 1-50.
Hirschfield, RM (2001) Bipolar spectrum disorders. J Clin Psychiatry. 62 (Suppl 14): 5-9.
Judd, L., Akiskal, H.S., et al. (2002) The long term natural history of the
weekly symptomatic status of bipolar I disorder. Archives of Psychiatry. 59:
530-537
Katz, S.J., et al. (1998) Medication management of depression in the U.S. and Ontario. J. Gen. Inter. Med.13: 77-85.
Katz, S.J., Kessler, R.C., Lin, E., Wells, K.B. (1998) Medication management of depression in the U.S. and Ontario. Journal of General Internal Medicine. 13: 77-85.
Kessler, R.C., McGonagle, K.A., Zhao, S., Nelson, C.B., Hughes, M., Eshleman, S., Wittchen, H.U., and Kendler, K.S. (1994) Lifetime and 12-month prevalence of DSM-IV psychiatric disorders in the United States. Results from the National Comorbidity Survey. Archives of General Psychiatry, 151 (1): 8-19.
Kuczenski, R., et al. (2002) Exposure of adolescent rats to oral methylphenidate. J. Neurosci. 22:264-271. (No evidence of addiction).
Lawlor, D.A., and Hopker, S.W. (2001) The effectiveness of exercise on an intervention in the management of depression: a systematic review and meta-regression analysis of randomized controlled trials. British Medical Journal. 322 (7289): 263-767.
Linde, K., et al. (2008) St. John's Wort for Depression. Cochrane Database
of Systematic Reviews. Issue 4. No. CD000448.DOI : 10.1002/14651858.CD000448.pub3
Le Doux (2015) Anxious. Penguin: New York.
Lu, S.C. (2000) S-Adenosylmethionine (SAM-e). International Journal of Biochemistry and Cell Biology. 32, 391-395.
Madhukar, H., et al. (2001) Algorithm for the treatment of chronic depression. J. Clin Psychiatry. 62 (Suppl 6): 22-29.
Madhukar, H., et al. (2001) Using Treatment algorithms for the effective management of treatment-resistant depression. J Clin Psychiatry. 62 (Suppl 18): 25-29.
Malkoff-Schwartz, S., Frank, E. et al. (1998) Stressful life events and social rhythm disruption in the onset of manic depressive bipolar episodes: A preliminary investigation. Archives of Gen. Psych., 55, 702-707.
McCullough, J.P. (2000) Treatment for Chronic Depression. The Guilford Press, New York.
Metzner, R.J. (2000) Neurotransmitters and depression: New Possibilities. Annual Meeting: American Psychiatric Association.
MTA Cooperative Group (1999) A 14-month randomized clinical trial of treatment strategies for ADHD. Archives of General Psychiatry. 56(12), 1073-1086.
Murray, C.J.L., and Lopez, A.D., (Editors) (1996) Global burden of disease, Vol. 1 Geneva: Harvard School of Public Health on Behalf of the World Health Organization and the World Bank.
Nierenberg, A.A., Keefe, B.R., Leslie V.C., et al. (1999) Residual symptoms in depressed patients who respond acutely to fluoxetine. J Clin Psychiatry. 60: 221-225.
NIMH (2002) Sequenced Treatment Alternatives to Relieve Depression: www.edc.gsph.pitt.edu/stard
Nolen, W.A., et al. (2000) Treatment of bipolar depression, a review of the literature and suggestion for an algorithm. Neuropsychobio. 42, (suppl 1), 11-17.
Nutt, D.J. (2002) The neuropharmacology of serotonin and noradrenaline in depression. International Clinical Psychopharmacology, 17 (Suppl 1): 51-512.
Olfson, M. et al. (2002) National trends in the outpatient treatment of depression. JAMA. 287 (2): 203-209
Papolos, D. and Papolos, J. (1999) The Bi-Polar Child, Broadway Books: New York.
Paykel, E.S., Ramana, R., Cooper, Z., Hayhurst, H., Kerr, J., and Barocka, A. (1995) Residual symptoms after partial remission: An important outcome in depression. Psychological Medicine, 25: 1171-1180.
Peet, M. and Horrobin, D.F. (2002) A dose ranging study of the effects of ethyl-eicosapentaenoate in patients with ongoing depression despite apparently adequate treatment with standard drugs. Archives of General Psychiatry. 59: 913-919.
Phelps, J. (2016) A Spectrum Approach to Mood Disorders. WW Norton: Ney York.
Pope, M, and Scott, J. (2003) Do clinicians understand why individuals stop taking lithium? Journal of Affective Disorders. 74: 287-291.
Power, Mick (editor) (2004) Mood Disorders: A Handbook of Science and Practice. W. Sussex, England: John Wiley and Sons
Preston, J.D. (2006) Integrative Brief Therapy; Second edition. Impact Publishers,
Atascadero, CA
Preston, J.D. et al. (2017) Handbook of Clinical Psychopharmacology for Therapists;
Eighth Edition. New Harbinger, Oakland.
Reynolds, C.F., Frank, E., Perel, J.M., Imber, S.D., Cornes, C., Miller, M.D., Mazumdar, S.,Houck, P.R., Dew, M.A., Stack, J.A., Pollack, B.G., Kupfer, D.J. (1999) Nortriptyline and interpersonal psychotherapy as maintenance therapies for recurrent major depression: a randomized controlled trial in patients older than 59 years. JAMA. 281: 39-45
Riddle, S. (2001) Drug interactions: Examining the impact of botanicals and dietary supplements. Future Dimensions in Clinical Nutrition Management. 20:3, 7-14.
Rosenthal, N, (2013) Winter Blues Survival Guide. Guilford Press: New York
Schatzberg, A.F and DeBattista, C., (2002) Current psychotropic dosing and monitoring guidelines: 2002. Primary Psychiatry. 9 (6), 34-48.
Shelton, R.C. and Tomarken, A.J. (2001) Can recovery from depression be achieved? Psychiatric Services. 52 (11): 1469-1478.
Spiegel, D.A. and Bruce, T.J. (1997) Benzodiazepines and exposure-based cognitive behavior therapies for panic disorder: Conclusions from combined treatment trials. American Journal of Psychiatry, 154: 773-781.
Stahl, S. (2013) Essential Psychopharmacology. Cambridge University Press, Cambridge.
Stoll, A.L., Severus, W.E., Freeman, M.P., et al. (1999) Omega 3 fatty acids in bipolar disorder. A preliminary double-blind, placebo-controlled trial. Arch Gen Psychiatry, 56: 407-412.
Terman, M, Terman, J.S., and Ross, D.C. (1998) A controlled trial of timed bright light and negative ionization for treatment of winter depression. Archives of General Psychiatry. 55:875-882.
Texas Dept. of Mental Health (1998) Texas Medication Algorithm Project. Dallas, Texas.
Thase, M.E., Rush A.J. (1997) When At First You Don't Succeed: Sequential Strategies for Antidepressant Nonresponders. J Clin Psychiatry. 58 (Suppl 13): 23-29, PRPsy 22-3 (1A), April 1998.
Tohen, M., Baker, R.A. (2004) A placebo-controlled trial of olanzapine and olanzapine-fluoxetine combination in bipolar depression. Arch Gen Psychiatry. In press.
Trivedi, M.H., and Klieber, B.A., (2001) Algorithm for the treatment of chronic depression. J Clin Psychiatry. 62 (Suppl 6): 22-29.
Ustun, T.B., et al. (1995) In: Mental Illness in General Health Care: An International Study. John Wiley & Sons, London.
Wells, K.B. et al. (1994) Use of minor tranquilizers and antidepressant medications by depressed patients. American Journal of Psychiatry. 151 (5), 694-700.
Whiskey, E., et al. (2001) A systematic review and meta-analysis of hypericum perforatum in depression: a comprehensive clinical review. Inter. Clin. Psychopharmacology. 16: 239-252.
Wisner, K.L., et al. (2002) Verapamil treatment for women with bipolar disorder. Biol. Psychiatry. 51: 745-752.
Zimmerman, M., Posternak, M.A., and Chelminski, I. (2002) Symptom severity and exclusion from antidepressant efficacy trials. Journal of Clinical Psychopharmacology. 22 (6): 610- 614.
References: Neurobiology
Arborelius, L., et al. (1999) The role of cortico-tropin releasing factor in depression and anxiety disorders. Journal of Endocrinology, 166, p. 1-12.
Brown, E.S., et al. (1999) Hippocampal remodeling and damage by corticosteroids: Implications for mood disorders. Neuropsychopharmacology, 21. p. 474-484.
Gorman, J.M. et al. (2000) Neuroanatomical hypothesis of panic disorder, revised. American Journal of Psychiatry. 157(4): 493-505.
Kempermann G. and Gage, F.H. (1999) New nerve cells for the adult brain. Scientific American, Vol. 280, No. 5, p. 48-53.
Lawlor, D.A., Hopker, S.W. (2001) The effectiveness of exercise as an intervention in the management of depression: systematic review and meta-regression analysis of randomized controlled trials. BMJ. 322 (7289): 763-767. Department of Social Medicine, University of Bristol, Bristol BS8 2PR, UK.
LeDoux, J. (2016) Anxious. Norton: New York.
Nestler, E.J., et al. (2001) Molecular Neuropharmacology. New York, McGraw-Hill.
Sapolsky, R.M. (2017) Behave. Penguin Random House: New York.


 ContinuingEdCourses.Net is approved by the American Psychological Association (APA) to sponsor continuing education for psychologists. ContinuingEdCourses.Net maintains responsibility for this program and its content.
ContinuingEdCourses.Net is approved by the American Psychological Association (APA) to sponsor continuing education for psychologists. ContinuingEdCourses.Net maintains responsibility for this program and its content. ContinuingEdCourses.Net, provider #1107, is approved as an ACE provider to offer social work continuing education by the Association of Social Work Boards (ASWB) Approved Continuing Education (ACE) program. Regulatory boards are the final authority on courses accepted for continuing education credit. ACE provider approval period: 3/9/2005-3/9/2027.
ContinuingEdCourses.Net, provider #1107, is approved as an ACE provider to offer social work continuing education by the Association of Social Work Boards (ASWB) Approved Continuing Education (ACE) program. Regulatory boards are the final authority on courses accepted for continuing education credit. ACE provider approval period: 3/9/2005-3/9/2027. ContinuingEdCourses.Net has been approved by the National Board for Certified Counselors (NBCC) as an Approved Continuing Education Provider (ACEP), ACEP #6323. Programs that do not qualify for NBCC credit are clearly identified. ContinuingEdCourses.Net is solely responsible for all aspects of the programs.
ContinuingEdCourses.Net has been approved by the National Board for Certified Counselors (NBCC) as an Approved Continuing Education Provider (ACEP), ACEP #6323. Programs that do not qualify for NBCC credit are clearly identified. ContinuingEdCourses.Net is solely responsible for all aspects of the programs. ContinuingEdCourses.Net is recognized by the New York State Education Department's State Board for Psychology (NYSED-PSY) as an approved provider of continuing education for licensed psychologists #PSY-0048.
ContinuingEdCourses.Net is recognized by the New York State Education Department's State Board for Psychology (NYSED-PSY) as an approved provider of continuing education for licensed psychologists #PSY-0048.
